| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M4 |
|---|
| Ligand | BDBM39341 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_139254 |
|---|
| Ki | 330±n/a nM |
|---|
| Citation |  Karton, Y; Bradbury, BJ; Baumgold, J; Paek, R; Jacobson, KA Functionalized congener approach to muscarinic antagonists: analogues of pirenzepine. J Med Chem34:2133-45 (1991) [PubMed] Karton, Y; Bradbury, BJ; Baumgold, J; Paek, R; Jacobson, KA Functionalized congener approach to muscarinic antagonists: analogues of pirenzepine. J Med Chem34:2133-45 (1991) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Muscarinic acetylcholine receptor M4 |
|---|
| Name: | Muscarinic acetylcholine receptor M4 |
|---|
| Synonyms: | ACM4_RAT | Cholinergic, muscarinic M4 | Chrm-4 | Chrm4 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 52841.70 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholinergic, muscarinic M4 CHRM4 RAT::P08485 |
|---|
| Residue: | 477 |
|---|
| Sequence: | MNFTPVNGSSANQSVRLVTAAHNHLETVEMVFIATVTGSLSLVTVVGNILVMLSIKVNRQ
LQTVNNYFLFSLGCADLIIGAFSMNLYTLYIIKGYWPLGAVVCDLWLALDYVVSNASVMN
LLIISFDRYFCVTKPLTYPARRTTKMAGLMIAAAWVLSFVLWAPAILFWQFVVGKRTVPD
NQCFIQFLSNPAVTFGTAIAAFYLPVVIMTVLYIHISLASRSRVHKHRPEGPKEKKAKTL
AFLKSPLMKPSIKKPPPGGASREELRNGKLEEAPPPALPPPPRPVPDKDTSNESSSGSAT
QNTKERPPTELSTAEATTPALPAPTLQPRTLNPASKWSKIQIVTKQTGNECVTAIEIVPA
TPAGMRPAANVARKFASIARNQVRKKRQMAARERKVTRTIFAILLAFILTWTPYNVMVLV
NTFCQSCIPERVWSIGYWLCYVNSTINPACYALCNATFKKTFRHLLLCQYRNIGTAR
|
|
|
|---|
| BDBM39341 |
|---|
| n/a |
|---|
| Name | BDBM39341 |
|---|
| Synonyms: | 11-[(4-methylpiperazin-1-yl)acetyl]-5,11-dihydro-6H-pyrido[2,3-b][1,4]benzodiazepin-6-one | 11-[2-(4-methyl-1-piperazinyl)-1-oxoethyl]-5H-pyrido[2,3-b][1,4]benzodiazepin-6-one;hydrochloride | 11-[2-(4-methylpiperazin-1-yl)acetyl]-5H-pyrido[2,3-b][1,4]benzodiazepin-6-one;hydrochloride | 11-[2-(4-methylpiperazin-1-yl)ethanoyl]-5H-pyrido[2,3-b][1,4]benzodiazepin-6-one;hydrochloride | 11-[2-(4-methylpiperazino)acetyl]-5H-pyrido[2,3-b][1,4]benzodiazepin-6-one;hydrochloride | CHEMBL9967 | MLS000069702 | PIRENZEPINE | PIRENZEPINE DIHYDROCHLORIDE | SMR000058502 | cid_185248 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H21N5O2 |
|---|
| Mol. Mass. | 351.4023 |
|---|
| SMILES | CN1CCN(CC(=O)N2c3ccccc3C(=O)Nc3cccnc23)CC1 |
|---|
| Structure | 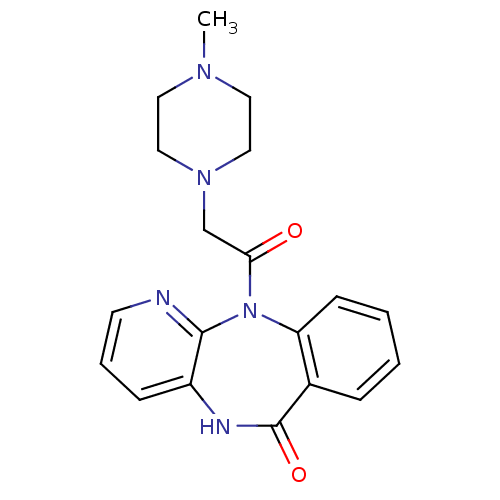
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Karton, Y; Bradbury, BJ; Baumgold, J; Paek, R; Jacobson, KA Functionalized congener approach to muscarinic antagonists: analogues of pirenzepine. J Med Chem34:2133-45 (1991) [PubMed]
Karton, Y; Bradbury, BJ; Baumgold, J; Paek, R; Jacobson, KA Functionalized congener approach to muscarinic antagonists: analogues of pirenzepine. J Med Chem34:2133-45 (1991) [PubMed]