| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Serine/threonine-protein kinase haspin |
|---|
| Ligand | BDBM50256734 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1714293 (CHEMBL4124342) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Dao, VH; Ourliac-Garnier, I; Bazin, MA; Jacquot, C; Baratte, B; Ruchaud, S; Bach, S; Grovel, O; Le Pape, P; Marchand, P Benzofuro[3,2-d]pyrimidines inspired from cercosporamide CaPkc1 inhibitor: Synthesis and evaluation of fluconazole susceptibility restoration. Bioorg Med Chem Lett28:2250-2255 (2018) [PubMed] Article Dao, VH; Ourliac-Garnier, I; Bazin, MA; Jacquot, C; Baratte, B; Ruchaud, S; Bach, S; Grovel, O; Le Pape, P; Marchand, P Benzofuro[3,2-d]pyrimidines inspired from cercosporamide CaPkc1 inhibitor: Synthesis and evaluation of fluconazole susceptibility restoration. Bioorg Med Chem Lett28:2250-2255 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Serine/threonine-protein kinase haspin |
|---|
| Name: | Serine/threonine-protein kinase haspin |
|---|
| Synonyms: | GSG2 | Germ cell-specific gene 2 protein | H-haspin | HASPIN | HASP_HUMAN | Haploid germ cell-specific nuclear protein kinase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 88531.28 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1510616 |
|---|
| Residue: | 798 |
|---|
| Sequence: | MAASLPGPGSRLFRTYGAADGRRQRRPGREAAQWFPPQDRRRFFNSSGSSDASIGDPSQS
DDPDDPDDPDFPGSPVRRRRRRPGGRVPKDRPSLTVTPKRWKLRARPSLTVTPRRLGLRA
RPPQKCSTPCGPLRLPPFPSRDSGRLSPDLSVCGQPRDGDELGISASLFSSLASPCPGSP
TPRDSVISIGTSACLVAASAVPSGLHLPEVSLDRASLPCSQEEATGGAKDTRMVHQTRAS
LRSVLFGLMNSGTPEDSEFRADGKNMRESCCKRKLVVGNGPEGPGLSSTGKRRATGQDSC
QERGLQEAVRREHQEASVPKGRIVPRGIDRLERTRSSRKSKHQEATETSLLHSHRFKKGQ
KLGKDSFPTQDLTPLQNVCFWTKTRASFSFHKKKIVTDVSEVCSIYTTATSLSGSLLSEC
SNRPVMNRTSGAPSSWHSSSMYLLSPLNTLSISNKKASDAEKVYGECSQKGPVPFSHCLP
TEKLQRCEKIGEGVFGEVFQTIADHTPVAIKIIAIEGPDLVNGSHQKTFEEILPEIIISK
ELSLLSGEVCNRTEGFIGLNSVHCVQGSYPPLLLKAWDHYNSTKGSANDRPDFFKDDQLF
IVLEFEFGGIDLEQMRTKLSSLATAKSILHQLTASLAVAEASLRFEHRDLHWGNVLLKKT
SLKKLHYTLNGKSSTIPSCGLQVSIIDYTLSRLERDGIVVFCDVSMDEDLFTGDGDYQFD
IYRLMKKENNNRWGEYHPYSNVLWLHYLTDKMLKQMTFKTKCNTPAMKQIKRKIQEFHRT
MLNFSSATDLLCQHSLFK
|
|
|
|---|
| BDBM50256734 |
|---|
| n/a |
|---|
| Name | BDBM50256734 |
|---|
| Synonyms: | (-)-Cercosporamide | CHEMBL475816 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H13NO7 |
|---|
| Mol. Mass. | 331.2769 |
|---|
| SMILES | CC(=O)C1C(=O)C=C2Oc3c(c(O)cc(O)c3C(N)=O)[C@]2(C)C1=O |r,t:6| |
|---|
| Structure | 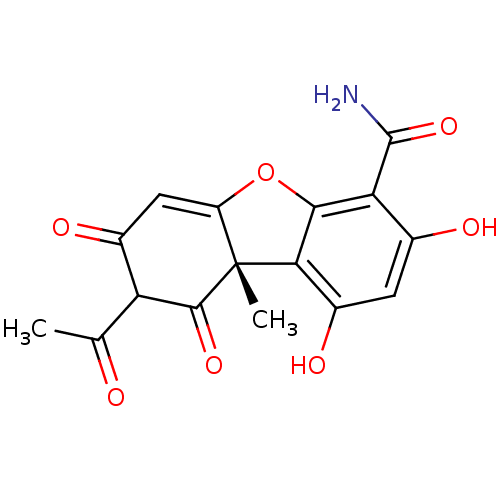
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dao, VH; Ourliac-Garnier, I; Bazin, MA; Jacquot, C; Baratte, B; Ruchaud, S; Bach, S; Grovel, O; Le Pape, P; Marchand, P Benzofuro[3,2-d]pyrimidines inspired from cercosporamide CaPkc1 inhibitor: Synthesis and evaluation of fluconazole susceptibility restoration. Bioorg Med Chem Lett28:2250-2255 (2018) [PubMed] Article
Dao, VH; Ourliac-Garnier, I; Bazin, MA; Jacquot, C; Baratte, B; Ruchaud, S; Bach, S; Grovel, O; Le Pape, P; Marchand, P Benzofuro[3,2-d]pyrimidines inspired from cercosporamide CaPkc1 inhibitor: Synthesis and evaluation of fluconazole susceptibility restoration. Bioorg Med Chem Lett28:2250-2255 (2018) [PubMed] Article