| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Activated CDC42 kinase 1 |
|---|
| Ligand | BDBM50242740 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1765761 (CHEMBL4201008) |
|---|
| IC50 | 18±n/a nM |
|---|
| Citation |  Cho, H; Shin, I; Ju, E; Choi, S; Hur, W; Kim, H; Hong, E; Kim, ND; Choi, HG; Gray, NS; Sim, T First SAR Study for Overriding NRAS Mutant Driven Acute Myeloid Leukemia. J Med Chem61:8353-8373 (2018) [PubMed] Article Cho, H; Shin, I; Ju, E; Choi, S; Hur, W; Kim, H; Hong, E; Kim, ND; Choi, HG; Gray, NS; Sim, T First SAR Study for Overriding NRAS Mutant Driven Acute Myeloid Leukemia. J Med Chem61:8353-8373 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Activated CDC42 kinase 1 |
|---|
| Name: | Activated CDC42 kinase 1 |
|---|
| Synonyms: | ACK1 | ACK1_HUMAN | Activated CDC42 kinase 1 (ACK-1) | TNK2 | Tyrosine kinase non-receptor protein 2 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 114577.86 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q07912 |
|---|
| Residue: | 1038 |
|---|
| Sequence: | MQPEEGTGWLLELLSEVQLQQYFLRLRDDLNVTRLSHFEYVKNEDLEKIGMGRPGQRRLW
EAVKRRKALCKRKSWMSKVFSGKRLEAEFPPHHSQSTFRKTSPAPGGPAGEGPLQSLTCL
IGEKDLRLLEKLGDGSFGVVRRGEWDAPSGKTVSVAVKCLKPDVLSQPEAMDDFIREVNA
MHSLDHRNLIRLYGVVLTPPMKMVTELAPLGSLLDRLRKHQGHFLLGTLSRYAVQVAEGM
GYLESKRFIHRDLAARNLLLATRDLVKIGDFGLMRALPQNDDHYVMQEHRKVPFAWCAPE
SLKTRTFSHASDTWMFGVTLWEMFTYGQEPWIGLNGSQILHKIDKEGERLPRPEDCPQDI
YNVMVQCWAHKPEDRPTFVALRDFLLEAQPTDMRALQDFEEPDKLHIQMNDVITVIEGRA
ENYWWRGQNTRTLCVGPFPRNVVTSVAGLSAQDISQPLQNSFIHTGHGDSDPRHCWGFPD
RIDELYLGNPMDPPDLLSVELSTSRPPQHLGGVKKPTYDPVSEDQDPLSSDFKRLGLRKP
GLPRGLWLAKPSARVPGTKASRGSGAEVTLIDFGEEPVVPALRPCAPSLAQLAMDACSLL
DETPPQSPTRALPRPLHPTPVVDWDARPLPPPPAYDDVAQDEDDFEICSINSTLVGAGVP
AGPSQGQTNYAFVPEQARPPPPLEDNLFLPPQGGGKPPSSAQTAEIFQALQQECMRQLQA
PAGSPAPSPSPGGDDKPQVPPRVPIPPRPTRPHVQLSPAPPGEEETSQWPGPASPPRVPP
REPLSPQGSRTPSPLVPPGSSPLPPRLSSSPGKTMPTTQSFASDPKYATPQVIQAPGPRA
GPCILPIVRDGKKVSSTHYYLLPERPSYLERYQRFLREAQSPEEPTPLPVPLLLPPPSTP
APAAPTATVRPMPQAALDPKANFSTNNSNPGARPPPPRATARLPQRGCPGDGPEAGRPAD
KIQMAMVHGVTTEECQAALQCHGWSVQRAAQYLKVEQLFGLGLRPRGECHKVLEMFDWNL
EQAGCHLLGSWGPAHHKR
|
|
|
|---|
| BDBM50242740 |
|---|
| n/a |
|---|
| Name | BDBM50242740 |
|---|
| Synonyms: | CHEMBL459850 | N-(3-(7-Amino-1-methyl-2-oxo-1,2-dihydropyrimido[4,5-d]pyrimidin-3(4H)-yl)-4-methylphenyl)-3-(trifluoromethyl)benzamide | Type II inhibitor, 14 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H19F3N6O2 |
|---|
| Mol. Mass. | 456.4205 |
|---|
| SMILES | CN1C(=O)N(Cc2cnc(N)nc12)c1cc(NC(=O)c2cccc(c2)C(F)(F)F)ccc1C |
|---|
| Structure | 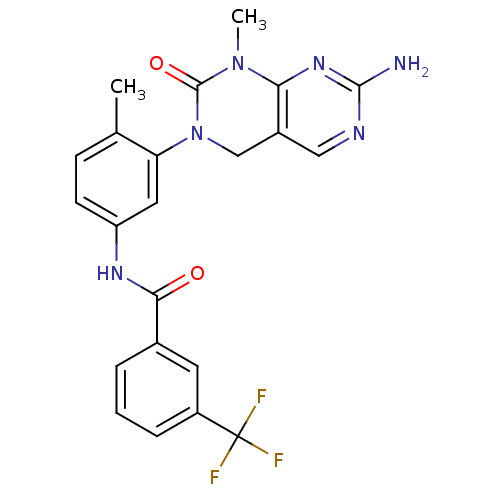
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cho, H; Shin, I; Ju, E; Choi, S; Hur, W; Kim, H; Hong, E; Kim, ND; Choi, HG; Gray, NS; Sim, T First SAR Study for Overriding NRAS Mutant Driven Acute Myeloid Leukemia. J Med Chem61:8353-8373 (2018) [PubMed] Article
Cho, H; Shin, I; Ju, E; Choi, S; Hur, W; Kim, H; Hong, E; Kim, ND; Choi, HG; Gray, NS; Sim, T First SAR Study for Overriding NRAS Mutant Driven Acute Myeloid Leukemia. J Med Chem61:8353-8373 (2018) [PubMed] Article