| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50218197 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1798022 (CHEMBL4270139) |
|---|
| Ki | 267±n/a nM |
|---|
| Citation |  Gogoi, D; Pal, A; Chattopadhyay, P; Paul, S; Deka, RC; Mukherjee, AK First Report of Plant-Derived ?-Sitosterol with Antithrombotic, in Vivo Anticoagulant, and Thrombus-Preventing Activities in a Mouse Model. J Nat Prod81:2521-2530 (2018) [PubMed] Article Gogoi, D; Pal, A; Chattopadhyay, P; Paul, S; Deka, RC; Mukherjee, AK First Report of Plant-Derived ?-Sitosterol with Antithrombotic, in Vivo Anticoagulant, and Thrombus-Preventing Activities in a Mouse Model. J Nat Prod81:2521-2530 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50218197 |
|---|
| n/a |
|---|
| Name | BDBM50218197 |
|---|
| Synonyms: | (-)-beta-Sitosterol | (24R)-Ethylcholest-5-en-3beta-ol | (24R)-Stigmast-5-en-3beta-ol | (3beta)-Stigmast-5-en-3-ol | 22,23-Dihydrostigmasterol | 24alpha-Ethylcholesterol | Azuprostat | CHEMBL221542 | Cupreol | Nimbosterol | Triastonal | alpha-Dihydrofucosterol | beta-Sitosterin | sitosterol | stigmast-5-en-3beta-ol |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H50O |
|---|
| Mol. Mass. | 414.7067 |
|---|
| SMILES | CC[C@H](CC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3CC=C4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@]12C)C(C)C |r,t:13| |
|---|
| Structure | 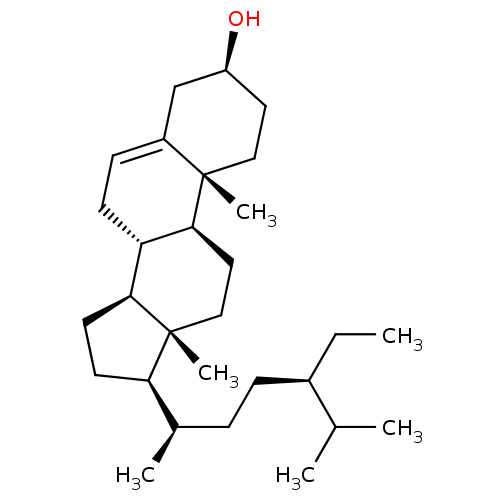
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gogoi, D; Pal, A; Chattopadhyay, P; Paul, S; Deka, RC; Mukherjee, AK First Report of Plant-Derived ?-Sitosterol with Antithrombotic, in Vivo Anticoagulant, and Thrombus-Preventing Activities in a Mouse Model. J Nat Prod81:2521-2530 (2018) [PubMed] Article
Gogoi, D; Pal, A; Chattopadhyay, P; Paul, S; Deka, RC; Mukherjee, AK First Report of Plant-Derived ?-Sitosterol with Antithrombotic, in Vivo Anticoagulant, and Thrombus-Preventing Activities in a Mouse Model. J Nat Prod81:2521-2530 (2018) [PubMed] Article