| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Type-1 angiotensin II receptor B |
|---|
| Ligand | BDBM50006909 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_36637 (CHEMBL652348) |
|---|
| IC50 | 7.5±n/a nM |
|---|
| Citation |  Mederski, WW; Dorsch, D; Bokel, HH; Beier, N; Lues, I; Schelling, P Non-peptide angiotensin II receptor antagonists: synthesis and biological activity of a series of novel 4,5-dihydro-4-oxo-3H-imidazo[4,5-c]pyridine derivatives. J Med Chem37:1632-45 (1994) [PubMed] Mederski, WW; Dorsch, D; Bokel, HH; Beier, N; Lues, I; Schelling, P Non-peptide angiotensin II receptor antagonists: synthesis and biological activity of a series of novel 4,5-dihydro-4-oxo-3H-imidazo[4,5-c]pyridine derivatives. J Med Chem37:1632-45 (1994) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Type-1 angiotensin II receptor B |
|---|
| Name: | Type-1 angiotensin II receptor B |
|---|
| Synonyms: | AGTRB_RAT | AT3 | Agtr1 | Agtr1b | Angiotensin II AT1B | Angiotensin II receptor (AT-1) type-1 | Angiotensin II type 1b (AT-1b) receptor | At1b | Type-1B angiotensin II receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 40929.44 |
|---|
| Organism: | RAT |
|---|
| Description: | Angiotensin II AT1B 0 RAT::P29089 |
|---|
| Residue: | 359 |
|---|
| Sequence: | MTLNSSTEDGIKRIQDDCPKAGRHNYIFVMIPTLYSIIFVVGIFGNSLVVIVIYFYMKLK
TVASVFLLNLALADLCFLLTLPLWAVYTAMEYRWPFGNHLCKIASASVSFNLYASVFLLT
CLSIDRYLAIVHPMKSRLRRTMLVAKVTCIIIWLMAGLASLPAVIYRNVYFIENTNITVC
AFHYESQNSTLPIGLGLTKNILGFVFPFLIILTSYTLIWKALKKAYKIQKNTPRNDDIFR
IIMAIVLFFFFSWVPHQIFTFLDVLIQLGIIRDCEIADIVDTAMPITICIAYFNNCLNPL
FYGFLGKKFKKYFLQLLKYIPPTAKSHAGLSTKMSTLSYRPSDNMSSSAKKSASFFEVE
|
|
|
|---|
| BDBM50006909 |
|---|
| n/a |
|---|
| Name | BDBM50006909 |
|---|
| Synonyms: | 2-Butyl-5-chloro-3-[2'-(1H-tetrazol-5-yl)-biphenyl-4-ylmethyl]-3H-imidazole-4-carboxylic acid | 2-Butyl-5-chloro-3-[2'-(1H-tetrazol-5-yl)-biphenyl-4-ylmethyl]-3H-imidazole-4-carboxylic acid (EXP3174) | 2-Butyl-5-chloro-3-[2'-(2H-tetrazol-5-yl)-biphenyl-4-ylmethyl]-3H-imidazole-4-carboxylic acid | 2-Butyl-5-chloro-3-[2'-(2H-tetrazol-5-yl)-biphenyl-4-ylmethyl]-3H-imidazole-4-carboxylic acid (EXP3174) | CHEMBL907 | Carboxylic Acid Metabolite (E-3174) | E 3174 | EXP3174 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H21ClN6O2 |
|---|
| Mol. Mass. | 436.894 |
|---|
| SMILES | CCCCc1nc(Cl)c(C(O)=O)n1Cc1ccc(cc1)-c1ccccc1-c1nnn[nH]1 |
|---|
| Structure | 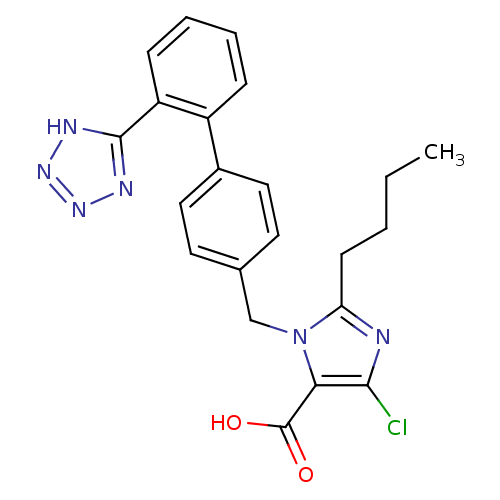
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mederski, WW; Dorsch, D; Bokel, HH; Beier, N; Lues, I; Schelling, P Non-peptide angiotensin II receptor antagonists: synthesis and biological activity of a series of novel 4,5-dihydro-4-oxo-3H-imidazo[4,5-c]pyridine derivatives. J Med Chem37:1632-45 (1994) [PubMed]
Mederski, WW; Dorsch, D; Bokel, HH; Beier, N; Lues, I; Schelling, P Non-peptide angiotensin II receptor antagonists: synthesis and biological activity of a series of novel 4,5-dihydro-4-oxo-3H-imidazo[4,5-c]pyridine derivatives. J Med Chem37:1632-45 (1994) [PubMed]