| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 60 kDa heat shock protein, mitochondrial |
|---|
| Ligand | BDBM46188 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1578874 (CHEMBL3813272) |
|---|
| IC50 | 7400±n/a nM |
|---|
| Citation |  Abdeen, S; Salim, N; Mammadova, N; Summers, CM; Frankson, R; Ambrose, AJ; Anderson, GG; Schultz, PG; Horwich, AL; Chapman, E; Johnson, SM GroEL/ES inhibitors as potential antibiotics. Bioorg Med Chem Lett26:3127-3134 (2016) [PubMed] Article Abdeen, S; Salim, N; Mammadova, N; Summers, CM; Frankson, R; Ambrose, AJ; Anderson, GG; Schultz, PG; Horwich, AL; Chapman, E; Johnson, SM GroEL/ES inhibitors as potential antibiotics. Bioorg Med Chem Lett26:3127-3134 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 60 kDa heat shock protein, mitochondrial |
|---|
| Name: | 60 kDa heat shock protein, mitochondrial |
|---|
| Synonyms: | 3.6.4.9 | 60 kDa chaperonin | 60 kDa heat shock protein, mitochondrial | CH60_HUMAN | CPN60 | Chaperonin 60 | HSP-60 | HSP60 | HSP60/HSP10 | HSPD1 | Heat shock protein 60 | HuCHA60 | Mitochondrial matrix protein P1 | P60 lymphocyte protein |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 61050.41 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_117701 |
|---|
| Residue: | 573 |
|---|
| Sequence: | MLRLPTVFRQMRPVSRVLAPHLTRAYAKDVKFGADARALMLQGVDLLADAVAVTMGPKGR
TVIIEQSWGSPKVTKDGVTVAKSIDLKDKYKNIGAKLVQDVANNTNEEAGDGTTTATVLA
RSIAKEGFEKISKGANPVEIRRGVMLAVDAVIAELKKQSKPVTTPEEIAQVATISANGDK
EIGNIISDAMKKVGRKGVITVKDGKTLNDELEIIEGMKFDRGYISPYFINTSKGQKCEFQ
DAYVLLSEKKISSIQSIVPALEIANAHRKPLVIIAEDVDGEALSTLVLNRLKVGLQVVAV
KAPGFGDNRKNQLKDMAIATGGAVFGEEGLTLNLEDVQPHDLGKVGEVIVTKDDAMLLKG
KGDKAQIEKRIQEIIEQLDVTTSEYEKEKLNERLAKLSDGVAVLKVGGTSDVEVNEKKDR
VTDALNATRAAVEEGIVLGGGCALLRCIPALDSLTPANEDQKIGIEIIKRTLKIPAMTIA
KNAGVEGSLIVEKIMQSSSEVGYDAMAGDFVNMVEKGIIDPTKVVRTALLDAAGVASLLT
TAEVVVTEIPKEEKDPGMGAMGGMGGGMGGGMF
|
|
|
|---|
| BDBM46188 |
|---|
| n/a |
|---|
| Name | BDBM46188 |
|---|
| Synonyms: | 1-[2,3-bis(2-furanyl)-6-quinoxalinyl]-3-methylurea | 1-[2,3-bis(2-furyl)quinoxalin-6-yl]-3-methyl-urea | 1-[2,3-bis(furan-2-yl)quinoxalin-6-yl]-3-methyl-urea | 1-[2,3-bis(furan-2-yl)quinoxalin-6-yl]-3-methylurea | MLS-0202054.0001 | cid_1092683 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H14N4O3 |
|---|
| Mol. Mass. | 334.3288 |
|---|
| SMILES | CNC(=O)Nc1ccc2nc(-c3ccco3)c(nc2c1)-c1ccco1 |
|---|
| Structure | 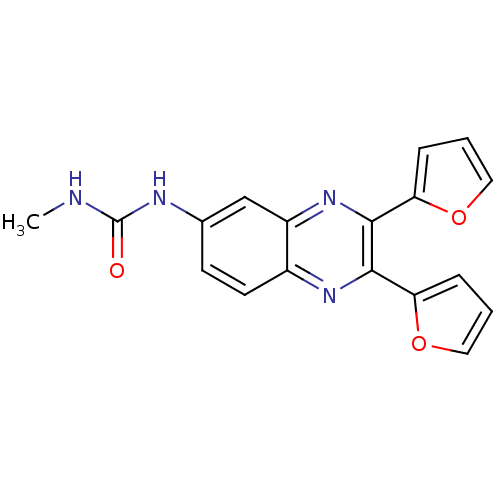
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Abdeen, S; Salim, N; Mammadova, N; Summers, CM; Frankson, R; Ambrose, AJ; Anderson, GG; Schultz, PG; Horwich, AL; Chapman, E; Johnson, SM GroEL/ES inhibitors as potential antibiotics. Bioorg Med Chem Lett26:3127-3134 (2016) [PubMed] Article
Abdeen, S; Salim, N; Mammadova, N; Summers, CM; Frankson, R; Ambrose, AJ; Anderson, GG; Schultz, PG; Horwich, AL; Chapman, E; Johnson, SM GroEL/ES inhibitors as potential antibiotics. Bioorg Med Chem Lett26:3127-3134 (2016) [PubMed] Article