

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 1A2 | ||
| Ligand | BDBM50503771 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1812127 (CHEMBL4311587) | ||
| IC50 | 689±n/a nM | ||
| Citation |  Kubo, M; Yamamoto, K; Itoh, T Design and synthesis of selective CYP1B1 inhibitor via dearomatization of ?-naphthoflavone. Bioorg Med Chem27:285-304 (2019) [PubMed] Article Kubo, M; Yamamoto, K; Itoh, T Design and synthesis of selective CYP1B1 inhibitor via dearomatization of ?-naphthoflavone. Bioorg Med Chem27:285-304 (2019) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 1A2 | |||
| Name: | Cytochrome P450 1A2 | ||
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 58423.38 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P05177 | ||
| Residue: | 516 | ||
| Sequence: |
| ||
| BDBM50503771 | |||
| n/a | |||
| Name | BDBM50503771 | ||
| Synonyms: | CHEMBL4476231 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C19H18O2S | ||
| Mol. Mass. | 310.41 | ||
| SMILES | S[C@H]1CC[C@@H](CC1)c1cc(=O)c2ccc3ccccc3c2o1 |r,wU:4.7,wD:1.0,(37.21,-3.23,;35.87,-3.99,;34.54,-3.21,;33.2,-3.97,;33.2,-5.51,;34.53,-6.29,;35.86,-5.53,;31.87,-6.27,;31.86,-7.82,;30.52,-8.59,;30.52,-10.13,;29.19,-7.82,;27.86,-8.6,;26.52,-7.82,;26.52,-6.28,;25.2,-5.52,;25.19,-3.99,;26.52,-3.21,;27.85,-3.98,;27.85,-5.51,;29.19,-6.27,;30.53,-5.49,)| | ||
| Structure | 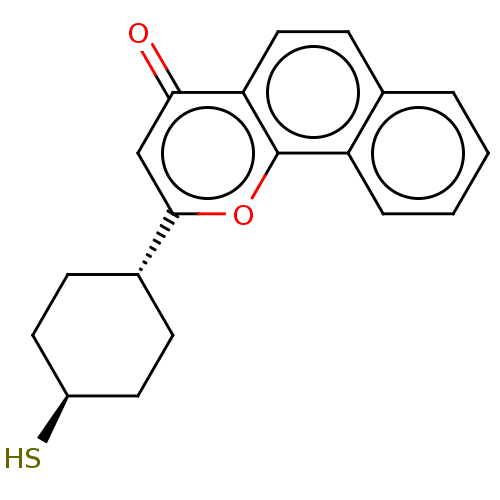
| ||