| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Indoleamine 2,3-dioxygenase 1 |
|---|
| Ligand | BDBM50126143 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1838980 (CHEMBL4339113) |
|---|
| IC50 | 7.9±n/a nM |
|---|
| Citation |  Steeneck, C; Kinzel, O; Anderhub, S; Hornberger, M; Pinto, S; Morschhaeuser, B; Braun, F; Kleymann, G; Hoffmann, T Discovery of Hydroxyamidine Based Inhibitors of IDO1 for Cancer Immunotherapy with Reduced Potential for Glucuronidation. ACS Med Chem Lett11:179-187 (2020) [PubMed] Article Steeneck, C; Kinzel, O; Anderhub, S; Hornberger, M; Pinto, S; Morschhaeuser, B; Braun, F; Kleymann, G; Hoffmann, T Discovery of Hydroxyamidine Based Inhibitors of IDO1 for Cancer Immunotherapy with Reduced Potential for Glucuronidation. ACS Med Chem Lett11:179-187 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Indoleamine 2,3-dioxygenase 1 |
|---|
| Name: | Indoleamine 2,3-dioxygenase 1 |
|---|
| Synonyms: | I23O1_HUMAN | IDO | IDO-1 | IDO1 | INDO | Indoleamine 2,3-Dioxygenasae (IDO) | Indoleamine 2,3-dioxygenase | Indoleamine-pyrrole 2,3-dioxygenase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 45330.80 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P14902 |
|---|
| Residue: | 403 |
|---|
| Sequence: | MAHAMENSWTISKEYHIDEEVGFALPNPQENLPDFYNDWMFIAKHLPDLIESGQLRERVE
KLNMLSIDHLTDHKSQRLARLVLGCITMAYVWGKGHGDVRKVLPRNIAVPYCQLSKKLEL
PPILVYADCVLANWKKKDPNKPLTYENMDVLFSFRDGDCSKGFFLVSLLVEIAAASAIKV
IPTVFKAMQMQERDTLLKALLEIASCLEKALQVFHQIHDHVNPKAFFSVLRIYLSGWKGN
PQLSDGLVYEGFWEDPKEFAGGSAGQSSVFQCFDVLLGIQQTAGGGHAAQFLQDMRRYMP
PAHRNFLCSLESNPSVREFVLSKGDAGLREAYDACVKALVSLRSYHLQIVTKYILIPASQ
QPKENKTSEDPSKLEAKGTGGTDLMNFLKTVRSTTEKSLLKEG
|
|
|
|---|
| BDBM50126143 |
|---|
| n/a |
|---|
| Name | BDBM50126143 |
|---|
| Synonyms: | Epacadostat | INCB-024360 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H13BrFN7O4S |
|---|
| Mol. Mass. | 438.233 |
|---|
| SMILES | NS(=O)(=O)NCCNc1nonc1\C(Nc1ccc(F)c(Br)c1)=N\O |
|---|
| Structure | 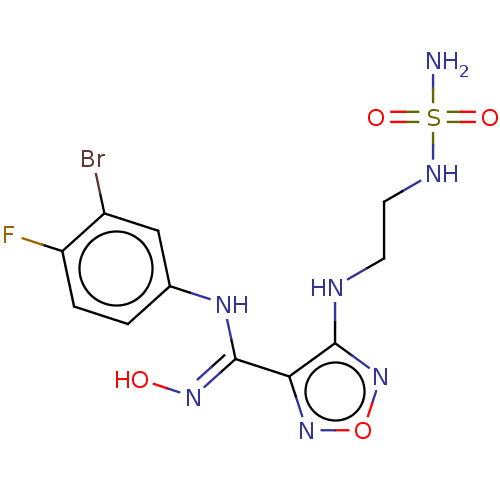
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Steeneck, C; Kinzel, O; Anderhub, S; Hornberger, M; Pinto, S; Morschhaeuser, B; Braun, F; Kleymann, G; Hoffmann, T Discovery of Hydroxyamidine Based Inhibitors of IDO1 for Cancer Immunotherapy with Reduced Potential for Glucuronidation. ACS Med Chem Lett11:179-187 (2020) [PubMed] Article
Steeneck, C; Kinzel, O; Anderhub, S; Hornberger, M; Pinto, S; Morschhaeuser, B; Braun, F; Kleymann, G; Hoffmann, T Discovery of Hydroxyamidine Based Inhibitors of IDO1 for Cancer Immunotherapy with Reduced Potential for Glucuronidation. ACS Med Chem Lett11:179-187 (2020) [PubMed] Article