| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Ligand | BDBM50000541 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1846642 (CHEMBL4347183) |
|---|
| IC50 | 830±n/a nM |
|---|
| Citation |  Li, Z; Wang, ZC; Li, X; Abbas, M; Wu, SY; Ren, SZ; Liu, QX; Liu, Y; Chen, PW; Duan, YT; Lv, PC; Zhu, HL Design, synthesis and evaluation of novel diaryl-1,5-diazoles derivatives bearing morpholine as potent dual COX-2/5-LOX inhibitors and antitumor agents. Eur J Med Chem169:168-184 (2019) [PubMed] Article Li, Z; Wang, ZC; Li, X; Abbas, M; Wu, SY; Ren, SZ; Liu, QX; Liu, Y; Chen, PW; Duan, YT; Lv, PC; Zhu, HL Design, synthesis and evaluation of novel diaryl-1,5-diazoles derivatives bearing morpholine as potent dual COX-2/5-LOX inhibitors and antitumor agents. Eur J Med Chem169:168-184 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Name: | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Synonyms: | Alox5 | Arachidonate 5-lipoxygenase | LOX5_RAT |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 78082.31 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1432947 |
|---|
| Residue: | 673 |
|---|
| Sequence: | MPSYTVTVATGSQWFAGTDDYIYLSLIGSAGCSEKHLLDKAFYNDFERGGRDSYDVTVDE
ELGEIYLVKIEKRKYRLHDDWYLKYITLKTPHDYIEFPCYRWITGEGEIVLRDGCAKLAR
DDQIHILKQHRRKELETRQKQYRWMEWNPGFPLSIDAKCHKDLPRDIQFDSEKGVDFVLN
YSKAMENLFINRFMHMFQSSWHDFADFEKIFVKISNTISERVKNHWQEDLMFGYQFLNGC
NPVLIKRCTELPKKLPVTTEMVECSLERQLSLEQEVQEGNIFIVDYELLDGIDANKTDPC
THQFLAAPICLLYKNLANKIVPIAIQLNQTPGEKNPIFLPTDSKYDWLLAKIWVRSSDFH
IHQTITHLLRTHLVSEVFGIAMYRQLPAVHPLFKLLVAHVRFTIAINTKAREQLNCEYGL
FDKANATGGGGHVQMVQRAVQDLTYSSLCFPEAIKARGMDNTEDIPYYFYRDDGLLVWEA
IQSFTTEVVSIYYEDDQVVEEDQELQDFVKDVYVYGMRGRKASGFPKSIKSREKLSEYLT
VVIFTASAQHAAVNFGQYDWCSWIPNAPPTMRAPPPTAKGVVTIEQIVDTLPDRGRSCWH
LGAVWALSQFQENELFLGMYPEEHFIEKPVKEAMIRFRKNLEAIVSVIAERNKNKKLPYY
YLSPDRIPNSVAI
|
|
|
|---|
| BDBM50000541 |
|---|
| n/a |
|---|
| Name | BDBM50000541 |
|---|
| Synonyms: | (+-)-1-(1-Benzo[b]thien-2-ylethyl)-1-hydroxyurea | 1-[1-(1-benzothien-2-yl)ethyl]-1-hydroxyurea | CHEMBL93 | Leutrol | N-(1-Benzo(b)thien-2-ylethyl)-N-hydroxyurea | N-[1-(benzo[b]thiophen-2-yl)ethyl]-N-hydroxyurea | ZILEUTON | Zyflo (TN) |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H12N2O2S |
|---|
| Mol. Mass. | 236.29 |
|---|
| SMILES | CC(N(O)C(N)=O)c1cc2ccccc2s1 |
|---|
| Structure | 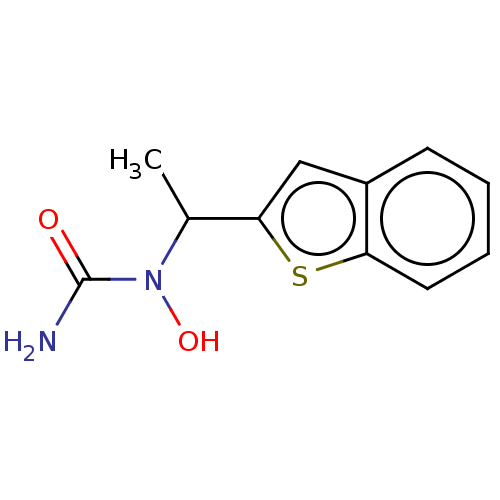
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, Z; Wang, ZC; Li, X; Abbas, M; Wu, SY; Ren, SZ; Liu, QX; Liu, Y; Chen, PW; Duan, YT; Lv, PC; Zhu, HL Design, synthesis and evaluation of novel diaryl-1,5-diazoles derivatives bearing morpholine as potent dual COX-2/5-LOX inhibitors and antitumor agents. Eur J Med Chem169:168-184 (2019) [PubMed] Article
Li, Z; Wang, ZC; Li, X; Abbas, M; Wu, SY; Ren, SZ; Liu, QX; Liu, Y; Chen, PW; Duan, YT; Lv, PC; Zhu, HL Design, synthesis and evaluation of novel diaryl-1,5-diazoles derivatives bearing morpholine as potent dual COX-2/5-LOX inhibitors and antitumor agents. Eur J Med Chem169:168-184 (2019) [PubMed] Article