| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Succinate dehydrogenase [ubiquinone] flavoprotein subunit, mitochondrial |
|---|
| Ligand | BDBM50116067 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1850349 (CHEMBL4350890) |
|---|
| IC50 | 8190±n/a nM |
|---|
| Citation |  Sun, AW; Bulterys, PL; Bartberger, MD; Jorth, PA; O'Boyle, BM; Virgil, SC; Miller, JF; Stoltz, BM Incorporation of a chiral gem-disubstituted nitrogen heterocycle yields an oxazolidinone antibiotic with reduced mitochondrial toxicity. Bioorg Med Chem Lett29:2686-2689 (2019) [PubMed] Article Sun, AW; Bulterys, PL; Bartberger, MD; Jorth, PA; O'Boyle, BM; Virgil, SC; Miller, JF; Stoltz, BM Incorporation of a chiral gem-disubstituted nitrogen heterocycle yields an oxazolidinone antibiotic with reduced mitochondrial toxicity. Bioorg Med Chem Lett29:2686-2689 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Succinate dehydrogenase [ubiquinone] flavoprotein subunit, mitochondrial |
|---|
| Name: | Succinate dehydrogenase [ubiquinone] flavoprotein subunit, mitochondrial |
|---|
| Synonyms: | 1.3.5.1 | Flavoprotein subunit of complex II | SDH2 | SDHA | SDHA_HUMAN | SDHF | Succinate dehydrogenase [ubiquinone] flavoprotein subunit, mitochondrial |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 72700.80 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_101537 |
|---|
| Residue: | 664 |
|---|
| Sequence: | MSGVRGLSRLLSARRLALAKAWPTVLQTGTRGFHFTVDGNKRASAKVSDSISAQYPVVDH
EFDAVVVGAGGAGLRAAFGLSEAGFNTACVTKLFPTRSHTVAAQGGINAALGNMEEDNWR
WHFYDTVKGSDWLGDQDAIHYMTEQAPAAVVELENYGMPFSRTEDGKIYQRAFGGQSLKF
GKGGQAHRCCCVADRTGHSLLHTLYGRSLRYDTSYFVEYFALDLLMENGECRGVIALCIE
DGSIHRIRAKNTVVATGGYGRTYFSCTSAHTSTGDGTAMITRAGLPCQDLEFVQFHPTGI
YGAGCLITEGCRGEGGILINSQGERFMERYAPVAKDLASRDVVSRSMTLEIREGRGCGPE
KDHVYLQLHHLPPEQLATRLPGISETAMIFAGVDVTKEPIPVLPTVHYNMGGIPTNYKGQ
VLRHVNGQDQIVPGLYACGEAACASVHGANRLGANSLLDLVVFGRACALSIEESCRPGDK
VPPIKPNAGEESVMNLDKLRFADGSIRTSELRLSMQKSMQNHAAVFRVGSVLQEGCGKIS
KLYGDLKHLKTFDRGMVWNTDLVETLELQNLMLCALQTIYGAEARKESRGAHAREDYKVR
IDEYDYSKPIQGQQKKPFEEHWRKHTLSYVDVGTGKVTLEYRPVIDKTLNEADCATVPPA
IRSY
|
|
|
|---|
| BDBM50116067 |
|---|
| n/a |
|---|
| Name | BDBM50116067 |
|---|
| Synonyms: | (Linezolid)N-[3-(3-Fluoro-4-morpholin-4-yl-phenyl)-2-oxo-oxazolidin-5-ylmethyl]-acetamide | (R)-N-((3-(3-fluoro-4-morpholinophenyl)-2-oxooxazolidin-5-yl)methyl)acetamide | (S)-3-(3-Fluoro-4-morpholin-4-yl-phenyl)-5-[(1-hydroxy-ethylamino)-methyl]-oxazolidin-2-one | (S)-N-((3-(3-fluoro-4-morpholinophenyl)-2-oxooxazolidin-5-yl)methyl)acetamide | (S)-N-((3-fluoro-4-morpholinophenyl)-2-oxaoxazolidin-5-yl)methyl)acetamide | Benzotriazol-2-yl-acetonitrile | CHEMBL126 | LINEZOLID | Linezoid | N-((S)-2-oxo-3-(S)-2,3,3a,4-tetrahydro-1H-benzo[b]pyrrolo[1,2-d][1,4]oxazin-7-yl-oxazolidin-5-ylmethyl)-acetamide | N-[(R)-3-(3-Fluoro-4-morpholin-4-yl-phenyl)-2-oxo-oxazolidin-5-ylmethyl]-acetamide | N-[(S)-3-(3-Fluoro-4-morpholin-4-yl-phenyl)-2-oxo-oxazolidin-5-ylmethyl]-acetamide | N-[3-(3-Fluoro-4-morpholin-4-yl-phenyl)-2-oxo-oxazolidin-5-ylmethyl]-acetamide | N-[3-(3-Fluoro-4-morpholin-4-yl-phenyl)-2-oxo-oxazolidin-5-ylmethyl]-acetamide(linezolid) | N-{[(5S)-3-(3-fluoro-4-morpholin-4-ylphenyl)-2-oxo-1,3-oxazolidin-5-yl]methyl}acetamide | U-100766 | Zyvox | cid_441401 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H20FN3O4 |
|---|
| Mol. Mass. | 337.3461 |
|---|
| SMILES | CC(=O)NC[C@H]1CN(C(=O)O1)c1ccc(N2CCOCC2)c(F)c1 |r| |
|---|
| Structure | 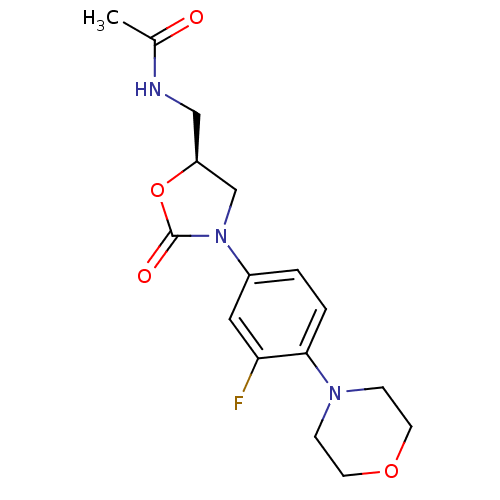
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sun, AW; Bulterys, PL; Bartberger, MD; Jorth, PA; O'Boyle, BM; Virgil, SC; Miller, JF; Stoltz, BM Incorporation of a chiral gem-disubstituted nitrogen heterocycle yields an oxazolidinone antibiotic with reduced mitochondrial toxicity. Bioorg Med Chem Lett29:2686-2689 (2019) [PubMed] Article
Sun, AW; Bulterys, PL; Bartberger, MD; Jorth, PA; O'Boyle, BM; Virgil, SC; Miller, JF; Stoltz, BM Incorporation of a chiral gem-disubstituted nitrogen heterocycle yields an oxazolidinone antibiotic with reduced mitochondrial toxicity. Bioorg Med Chem Lett29:2686-2689 (2019) [PubMed] Article