| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Indoleamine 2,3-dioxygenase 1 |
|---|
| Ligand | BDBM50514760 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1858297 (CHEMBL4359026) |
|---|
| IC50 | 9.3±n/a nM |
|---|
| Citation |  Serafini, M; Torre, E; Aprile, S; Grosso, ED; Ges�, A; Griglio, A; Colombo, G; Travelli, C; Paiella, S; Adamo, A; Orecchini, E; Coletti, A; Pallotta, MT; Ugel, S; Massarotti, A; Pirali, T; Fallarini, S Discovery of Highly Potent Benzimidazole Derivatives as Indoleamine 2,3-Dioxygenase-1 (IDO1) Inhibitors: From Structure-Based Virtual Screening to J Med Chem63:3047-3065 (2020) [PubMed] Article Serafini, M; Torre, E; Aprile, S; Grosso, ED; Ges�, A; Griglio, A; Colombo, G; Travelli, C; Paiella, S; Adamo, A; Orecchini, E; Coletti, A; Pallotta, MT; Ugel, S; Massarotti, A; Pirali, T; Fallarini, S Discovery of Highly Potent Benzimidazole Derivatives as Indoleamine 2,3-Dioxygenase-1 (IDO1) Inhibitors: From Structure-Based Virtual Screening to J Med Chem63:3047-3065 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Indoleamine 2,3-dioxygenase 1 |
|---|
| Name: | Indoleamine 2,3-dioxygenase 1 |
|---|
| Synonyms: | I23O1_MOUSE | IDO-1 | Ido | Ido1 | Indo | Indoleamine-pyrrole 2,3-dioxygenase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 45639.39 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_1452149 |
|---|
| Residue: | 407 |
|---|
| Sequence: | MALSKISPTEGSRRILEDHHIDEDVGFALPHPLVELPDAYSPWVLVARNLPVLIENGQLR
EEVEKLPTLSTDGLRGHRLQRLAHLALGYITMAYVWNRGDDDVRKVLPRNIAVPYCELSE
KLGLPPILSYADCVLANWKKKDPNGPMTYENMDILFSFPGGDCDKGFFLVSLLVEIAASP
AIKAIPTVSSAVERQDLKALEKALHDIATSLEKAKEIFKRMRDFVDPDTFFHVLRIYLSG
WKCSSKLPEGLLYEGVWDTPKMFSGGSAGQSSIFQSLDVLLGIKHEAGKESPAEFLQEMR
EYMPPAHRNFLFFLESAPPVREFVISRHNEDLTKAYNECVNGLVSVRKFHLAIVDTYIMK
PSKKKPTDGDKSEEPSNVESRGTGGTNPMTFLRSVKDTTEKALLSWP
|
|
|
|---|
| BDBM50514760 |
|---|
| n/a |
|---|
| Name | BDBM50514760 |
|---|
| Synonyms: | CHEMBL4448402 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H20N4O |
|---|
| Mol. Mass. | 380.4418 |
|---|
| SMILES | O=C(NCc1cccc(Cn2cnc3ccccc23)c1)c1cc2ccccc2[nH]1 |
|---|
| Structure | 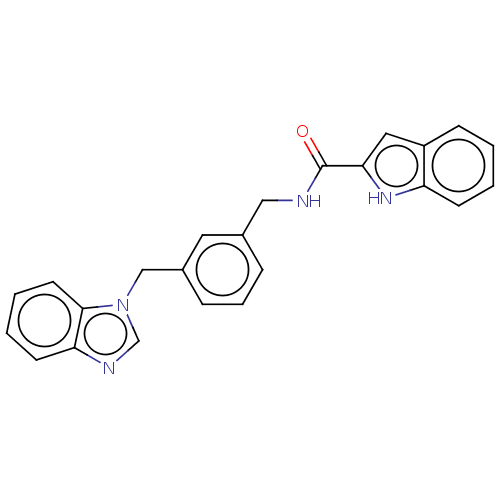
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Serafini, M; Torre, E; Aprile, S; Grosso, ED; Ges�, A; Griglio, A; Colombo, G; Travelli, C; Paiella, S; Adamo, A; Orecchini, E; Coletti, A; Pallotta, MT; Ugel, S; Massarotti, A; Pirali, T; Fallarini, S Discovery of Highly Potent Benzimidazole Derivatives as Indoleamine 2,3-Dioxygenase-1 (IDO1) Inhibitors: From Structure-Based Virtual Screening to J Med Chem63:3047-3065 (2020) [PubMed] Article
Serafini, M; Torre, E; Aprile, S; Grosso, ED; Ges�, A; Griglio, A; Colombo, G; Travelli, C; Paiella, S; Adamo, A; Orecchini, E; Coletti, A; Pallotta, MT; Ugel, S; Massarotti, A; Pirali, T; Fallarini, S Discovery of Highly Potent Benzimidazole Derivatives as Indoleamine 2,3-Dioxygenase-1 (IDO1) Inhibitors: From Structure-Based Virtual Screening to J Med Chem63:3047-3065 (2020) [PubMed] Article