| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protease |
|---|
| Ligand | BDBM50520742 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1882113 (CHEMBL4383612) |
|---|
| Ki | 0.140000±n/a nM |
|---|
| Citation |  Ghosh, AK; Jadhav, RD; Simpson, H; Kovela, S; Osswald, H; Agniswamy, J; Wang, YF; Hattori, SI; Weber, IT; Mitsuya, H Design, synthesis, and X-ray studies of potent HIV-1 protease inhibitors incorporating aminothiochromane and aminotetrahydronaphthalene carboxamide derivatives as the P2 ligands. Eur J Med Chem160:171-182 (2018) [PubMed] Article Ghosh, AK; Jadhav, RD; Simpson, H; Kovela, S; Osswald, H; Agniswamy, J; Wang, YF; Hattori, SI; Weber, IT; Mitsuya, H Design, synthesis, and X-ray studies of potent HIV-1 protease inhibitors incorporating aminothiochromane and aminotetrahydronaphthalene carboxamide derivatives as the P2 ligands. Eur J Med Chem160:171-182 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protease |
|---|
| Name: | Protease |
|---|
| Synonyms: | n/a |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 10904.79 |
|---|
| Organism: | Human immunodeficiency virus 1 (HIV-1) |
|---|
| Description: | Q9YQ12 |
|---|
| Residue: | 99 |
|---|
| Sequence: | PQITLWQRPFVTIKIEGQLKEALLDTGADDTVLEEMNLPGRWKPKMIGGIGGFIKVRQYD
QIVIEICGKKAIGTVLVGPTPVNIIGRNLLTQIGCTLNF
|
|
|
|---|
| BDBM50520742 |
|---|
| n/a |
|---|
| Name | BDBM50520742 |
|---|
| Synonyms: | CHEMBL4526105 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H49N3O8S |
|---|
| Mol. Mass. | 695.865 |
|---|
| SMILES | COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)c1ccc2[C@@H](O)CC[C@H](NC(=O)OC(C)(C)C)c2c1 |r| |
|---|
| Structure | 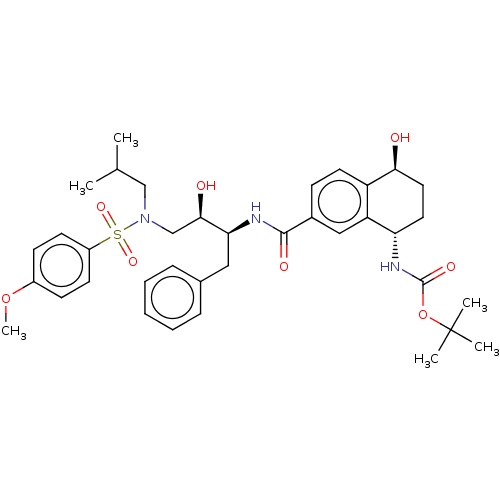
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ghosh, AK; Jadhav, RD; Simpson, H; Kovela, S; Osswald, H; Agniswamy, J; Wang, YF; Hattori, SI; Weber, IT; Mitsuya, H Design, synthesis, and X-ray studies of potent HIV-1 protease inhibitors incorporating aminothiochromane and aminotetrahydronaphthalene carboxamide derivatives as the P2 ligands. Eur J Med Chem160:171-182 (2018) [PubMed] Article
Ghosh, AK; Jadhav, RD; Simpson, H; Kovela, S; Osswald, H; Agniswamy, J; Wang, YF; Hattori, SI; Weber, IT; Mitsuya, H Design, synthesis, and X-ray studies of potent HIV-1 protease inhibitors incorporating aminothiochromane and aminotetrahydronaphthalene carboxamide derivatives as the P2 ligands. Eur J Med Chem160:171-182 (2018) [PubMed] Article