

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | 5-hydroxytryptamine receptor 1A | ||
| Ligand | BDBM50526023 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1898945 (CHEMBL4401060) | ||
| Ki | 218±n/a nM | ||
| Citation |  ?lifirski, G; Kr�l, M; Kleps, J; Ulenberg, S; Belka, M; B?czek, T; Siwek, A; Stachowicz, K; Szewczyk, B; Nowak, G; Bojarski, A; Kozio?, AE; Tur?o, J; Herold, F Synthesis of novel pyrido[1,2-c]pyrimidine derivatives with rigidized tryptamine moiety as potential SSRI and 5-HT Eur J Med Chem166:144-158 (2019) [PubMed] Article ?lifirski, G; Kr�l, M; Kleps, J; Ulenberg, S; Belka, M; B?czek, T; Siwek, A; Stachowicz, K; Szewczyk, B; Nowak, G; Bojarski, A; Kozio?, AE; Tur?o, J; Herold, F Synthesis of novel pyrido[1,2-c]pyrimidine derivatives with rigidized tryptamine moiety as potential SSRI and 5-HT Eur J Med Chem166:144-158 (2019) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| 5-hydroxytryptamine receptor 1A | |||
| Name: | 5-hydroxytryptamine receptor 1A | ||
| Synonyms: | 5-HT-1A | 5-HT1A | 5-hydroxytryptamine receptor 1A (5-HT-1A) | 5HT1A_HUMAN | ADRB2RL1 | ADRBRL1 | Dopamine D2 receptor and serotonin 1a receptor | G-21 | HTR1A | Serotonin receptor 1A | ||
| Type: | n/a | ||
| Mol. Mass.: | 46122.49 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | n/a | ||
| Residue: | 422 | ||
| Sequence: |
| ||
| BDBM50526023 | |||
| n/a | |||
| Name | BDBM50526023 | ||
| Synonyms: | CHEMBL4439370 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C31H30F2N4O2 | ||
| Mol. Mass. | 528.5923 | ||
| SMILES | Fc1ccc2[nH]cc(C3CCCN(CCCCn4c(=O)c(-c5ccccc5F)c5ccccn5c4=O)C3)c2c1 |(23.53,-42.07,;22.03,-42.37,;21.54,-43.84,;20.03,-44.14,;19.01,-42.99,;17.48,-42.98,;17.01,-41.51,;18.27,-40.62,;18.28,-39.08,;19.62,-38.32,;19.64,-36.78,;18.31,-35.99,;16.98,-36.76,;15.65,-35.98,;14.31,-36.75,;12.98,-35.97,;11.64,-36.73,;10.31,-35.95,;10.32,-34.41,;11.66,-33.65,;8.99,-33.63,;8.99,-32.1,;7.67,-31.33,;7.67,-29.79,;9.01,-29.02,;10.34,-29.79,;10.33,-31.33,;11.66,-32.11,;7.66,-34.4,;6.33,-33.62,;5,-34.4,;5,-35.94,;6.33,-36.7,;7.65,-35.94,;8.98,-36.71,;8.97,-38.25,;16.96,-38.3,;19.51,-41.53,;21.01,-41.23,)| | ||
| Structure | 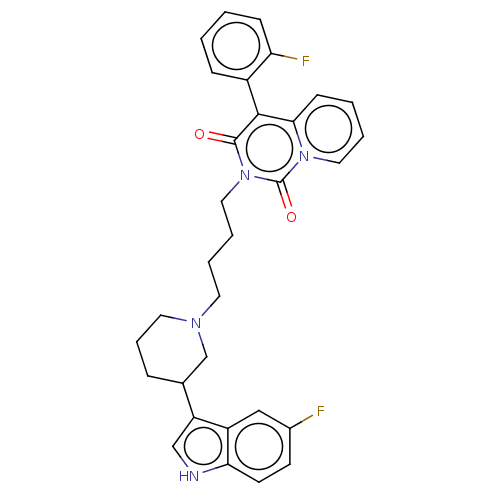
| ||