| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 4F3 |
|---|
| Ligand | BDBM50527963 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1904214 (CHEMBL4406436) |
|---|
| IC50 | 447000±n/a nM |
|---|
| Citation |  Kowalski, JP; McDonald, MG; Pelletier, RD; Hanenberg, H; Wiek, C; Rettie, AE Design and Characterization of the First Selective and Potent Mechanism-Based Inhibitor of Cytochrome P450 4Z1. J Med Chem63:4824-4836 (2020) [PubMed] Article Kowalski, JP; McDonald, MG; Pelletier, RD; Hanenberg, H; Wiek, C; Rettie, AE Design and Characterization of the First Selective and Potent Mechanism-Based Inhibitor of Cytochrome P450 4Z1. J Med Chem63:4824-4836 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 4F3 |
|---|
| Name: | Cytochrome P450 4F3 |
|---|
| Synonyms: | 1.14.14.1 | 1.14.14.79 | 1.14.14.94 | 20-HETE synthase | 20-hydroxyeicosatetraenoic acid synthase | CP4F3_HUMAN | CYP4F3 | CYPIVF3 | Cytochrome P450 4F3 | Cytochrome P450-LTB-omega | Docosahexaenoic acid omega-hydroxylase CYP4F3 | LTB4H | Leukotriene-B(4) 20-monooxygenase 2 | Leukotriene-B(4) omega-hydroxylase 2 | Synonyms=LTB4H |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 59856.15 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_119703 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MPQLSLSSLGLWPMAASPWLLLLLVGASWLLARILAWTYTFYDNCCRLRCFPQPPKRNWF
LGHLGLIHSSEEGLLYTQSLACTFGDMCCWWVGPWHAIVRIFHPTYIKPVLFAPAAIVPK
DKVFYSFLKPWLGDGLLLSAGEKWSRHRRMLTPAFHFNILKPYMKIFNESVNIMHAKWQL
LASEGSARLDMFEHISLMTLDSLQKCVFSFDSHCQEKPSEYIAAILELSALVTKRHQQIL
LYIDFLYYLTPDGQRFRRACRLVHDFTDAVIQERRRTLPSQGVDDFLQAKAKSKTLDFID
VLLLSKDEDGKKLSDEDIRAEADTFMFEGHDTTASGLSWVLYHLAKHPEYQERCRQEVQE
LLKDREPKEIEWDDLAQLPFLTMCIKESLRLHPPVPAVSRCCTQDIVLPDGRVIPKGIIC
LISVFGTHHNPAVWPDPEVYDPFRFDPKNIKERSPLAFIPFSAGPRNCIGQAFAMAEMKV
VLGLTLLRFRVLPDHTEPRRKPELVLRAEGGLWLRVEPLS
|
|
|
|---|
| BDBM50527963 |
|---|
| n/a |
|---|
| Name | BDBM50527963 |
|---|
| Synonyms: | CHEMBL4557940 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H20N4O2 |
|---|
| Mol. Mass. | 276.3342 |
|---|
| SMILES | OC(=O)CCCCCCCNn1nnc2ccccc12 |
|---|
| Structure | 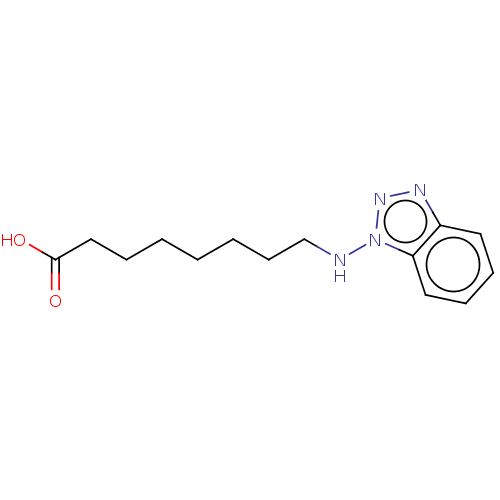
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kowalski, JP; McDonald, MG; Pelletier, RD; Hanenberg, H; Wiek, C; Rettie, AE Design and Characterization of the First Selective and Potent Mechanism-Based Inhibitor of Cytochrome P450 4Z1. J Med Chem63:4824-4836 (2020) [PubMed] Article
Kowalski, JP; McDonald, MG; Pelletier, RD; Hanenberg, H; Wiek, C; Rettie, AE Design and Characterization of the First Selective and Potent Mechanism-Based Inhibitor of Cytochrome P450 4Z1. J Med Chem63:4824-4836 (2020) [PubMed] Article