| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P2X purinoceptor 3 |
|---|
| Ligand | BDBM50532062 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1921835 (CHEMBL4424680) |
|---|
| IC50 | 8.0±n/a nM |
|---|
| Citation |  Tobinaga, H; Kameyama, T; Asahi, K; Horiguchi, T; Oohara, M; Tada, Y; Fuchino, K; Jikihara, S; Endoh, T; Kurihara, N; Kanda, Y; Ogawa, M; Tamura, N; Yagi, S; Taniguchi, E; Takahara, Y; Shimada, S; Takeyama, C; Yamamoto, S; Shinohara, S; Kai, H Pyrrolinone derivatives as a new class of P2X3 receptor antagonists Part 2: Discovery of orally bioavailable compounds. Bioorg Med Chem Lett29:688-693 (2019) [PubMed] Article Tobinaga, H; Kameyama, T; Asahi, K; Horiguchi, T; Oohara, M; Tada, Y; Fuchino, K; Jikihara, S; Endoh, T; Kurihara, N; Kanda, Y; Ogawa, M; Tamura, N; Yagi, S; Taniguchi, E; Takahara, Y; Shimada, S; Takeyama, C; Yamamoto, S; Shinohara, S; Kai, H Pyrrolinone derivatives as a new class of P2X3 receptor antagonists Part 2: Discovery of orally bioavailable compounds. Bioorg Med Chem Lett29:688-693 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P2X purinoceptor 3 |
|---|
| Name: | P2X purinoceptor 3 |
|---|
| Synonyms: | ATP receptor | Glucocorticoid receptor | P2RX3 | P2RX3_HUMAN | P2X purinoceptor 3 | P2X purinoceptor 3 (P2RX3) | P2X purinoceptor 3 (P2X3) | P2X3 | P2X3 purinoceptor | Purinergic receptor | p2x3 + hsa |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 44292.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P56373 |
|---|
| Residue: | 397 |
|---|
| Sequence: | MNCISDFFTYETTKSVVVKSWTIGIINRVVQLLIISYFVGWVFLHEKAYQVRDTAIESSV
VTKVKGSGLYANRVMDVSDYVTPPQGTSVFVIITKMIVTENQMQGFCPESEEKYRCVSDS
QCGPERLPGGGILTGRCVNYSSVLRTCEIQGWCPTEVDTVETPIMMEAENFTIFIKNSIR
FPLFNFEKGNLLPNLTARDMKTCRFHPDKDPFCPILRVGDVVKFAGQDFAKLARTGGVLG
IKIGWVCDLDKAWDQCIPKYSFTRLDSVSEKSSVSPGYNFRFAKYYKMENGSEYRTLLKA
FGIRFDVLVYGNAGKFNIIPTIISSVAAFTSVGVGTVLCDIILLNFLKGADQYKAKKFEE
VNETTLKIAALTNPVYPSDQTTAEKQSTDSGAFSIGH
|
|
|
|---|
| BDBM50532062 |
|---|
| n/a |
|---|
| Name | BDBM50532062 |
|---|
| Synonyms: | CHEMBL4439812 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H22NNaO4S |
|---|
| Mol. Mass. | 455.501 |
|---|
| SMILES | [Na;v0+].[#6]-[#8]-c1ccccc1-[#6@H]-1-[#7](-[#6](=O)-[#6](-[#8-])=[#6]-1-[#6](=O)-[#6](-[#6])-[#6])-c1ccc(cc1)-c1ccsc1 |r,c:14| |
|---|
| Structure | 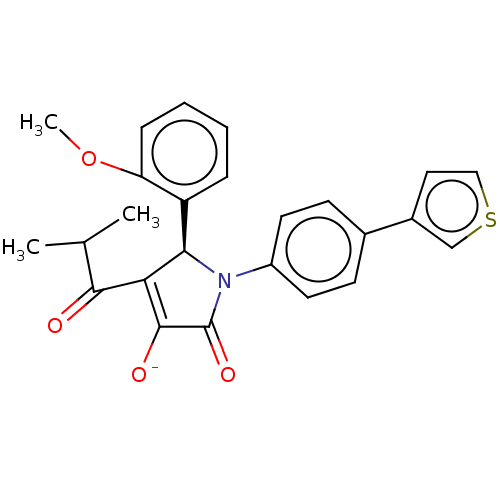
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tobinaga, H; Kameyama, T; Asahi, K; Horiguchi, T; Oohara, M; Tada, Y; Fuchino, K; Jikihara, S; Endoh, T; Kurihara, N; Kanda, Y; Ogawa, M; Tamura, N; Yagi, S; Taniguchi, E; Takahara, Y; Shimada, S; Takeyama, C; Yamamoto, S; Shinohara, S; Kai, H Pyrrolinone derivatives as a new class of P2X3 receptor antagonists Part 2: Discovery of orally bioavailable compounds. Bioorg Med Chem Lett29:688-693 (2019) [PubMed] Article
Tobinaga, H; Kameyama, T; Asahi, K; Horiguchi, T; Oohara, M; Tada, Y; Fuchino, K; Jikihara, S; Endoh, T; Kurihara, N; Kanda, Y; Ogawa, M; Tamura, N; Yagi, S; Taniguchi, E; Takahara, Y; Shimada, S; Takeyama, C; Yamamoto, S; Shinohara, S; Kai, H Pyrrolinone derivatives as a new class of P2X3 receptor antagonists Part 2: Discovery of orally bioavailable compounds. Bioorg Med Chem Lett29:688-693 (2019) [PubMed] Article