| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2C |
|---|
| Ligand | BDBM50544114 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1991783 (CHEMBL4625518) |
|---|
| IC50 | >3333±n/a nM |
|---|
| Citation |  Evans, CA; Zuluaga, A; Vasquez Matute, D; Baradaran-Noviri, S; Perez-Cervantes, N; Siegler, MA Synthesis and biological evaluation of thioadatanserin and its dialkylated products as partial 5-HTR Bioorg Med Chem Lett30:0 (2020) [PubMed] Article Evans, CA; Zuluaga, A; Vasquez Matute, D; Baradaran-Noviri, S; Perez-Cervantes, N; Siegler, MA Synthesis and biological evaluation of thioadatanserin and its dialkylated products as partial 5-HTR Bioorg Med Chem Lett30:0 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2C |
|---|
| Name: | 5-hydroxytryptamine receptor 2C |
|---|
| Synonyms: | 5-HT-1C | 5-HT-2C | 5-HT1C | 5-HT2C | 5-HT2C-INI | 5-HT2c VGI | 5-HTR2C | 5-hydroxytryptamine receptor 1C | 5-hydroxytryptamine receptor 2C (5-HT-2C) | 5-hydroxytryptamine receptor 2C (5HT-2C) | 5HT-1C | 5HT2C_HUMAN | HTR1C | HTR2C | Serotonin (5-HT3) receptor | Serotonin 2c (5-HT2c) receptor | Serotonin Receptor 2C |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 51836.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28335 |
|---|
| Residue: | 458 |
|---|
| Sequence: | MVNLRNAVHSFLVHLIGLLVWQSDISVSPVAAIVTDIFNTSDGGRFKFPDGVQNWPALSI
VIIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYVW
PLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVWA
ISIGVSVPIPVIGLRDEEKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYCLTIYV
LRRQALMLLHGHTEEPPGLSLDFLKCCKRNTAEEENSANPNQDQNARRRKKKERRPRGTM
QAINNERKASKVLGIVFFVFLIMWCPFFITNILSVLCEKSCNQKLMEKLLNVFVWIGYVC
SGINPLVYTLFNKIYRRAFSNYLRCNYKVEKKPPVRQIPRVAATALSGRELNVNIYRHTN
EPVIEKASDNEPGIEMQVENLELPVNPSSVVSERISSV
|
|
|
|---|
| BDBM50544114 |
|---|
| n/a |
|---|
| Name | BDBM50544114 |
|---|
| Synonyms: | CHEMBL4637409 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H31N5S |
|---|
| Mol. Mass. | 385.569 |
|---|
| SMILES | S=C(NCCN1CCN(CC1)c1ncccn1)C12CC3CC(CC(C3)C1)C2 |TLB:1:17:20:24.22.23,THB:22:21:18:24.23.25,22:23:20.21.26:18,25:23:20:26.17.18,25:17:20:24.22.23| |
|---|
| Structure | 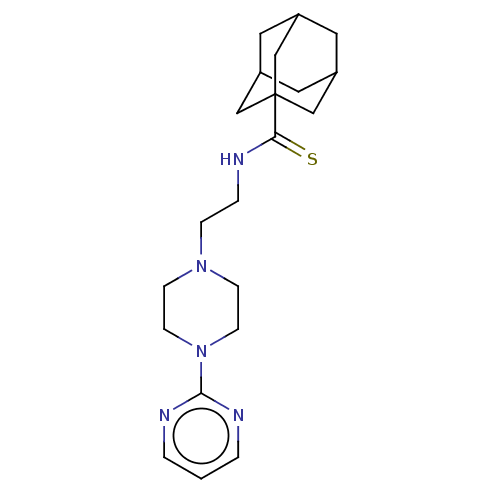
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Evans, CA; Zuluaga, A; Vasquez Matute, D; Baradaran-Noviri, S; Perez-Cervantes, N; Siegler, MA Synthesis and biological evaluation of thioadatanserin and its dialkylated products as partial 5-HTR Bioorg Med Chem Lett30:0 (2020) [PubMed] Article
Evans, CA; Zuluaga, A; Vasquez Matute, D; Baradaran-Noviri, S; Perez-Cervantes, N; Siegler, MA Synthesis and biological evaluation of thioadatanserin and its dialkylated products as partial 5-HTR Bioorg Med Chem Lett30:0 (2020) [PubMed] Article