

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Vasopressin V1a receptor | ||
| Ligand | BDBM50546419 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_2015644 (CHEMBL4669222) | ||
| IC50 | 500±n/a nM | ||
| Citation |  Boz�, �; Baska, F; L�vei, K; Sz�nt�, G; Dom�ny-Kov�cs, K; Kurk�, D; Szondin� Kord�s, K; Szokoli, T; Bata, I New V1a receptor antagonist. Part 2. Identification and optimization of triazolobenzazepines. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article Boz�, �; Baska, F; L�vei, K; Sz�nt�, G; Dom�ny-Kov�cs, K; Kurk�, D; Szondin� Kord�s, K; Szokoli, T; Bata, I New V1a receptor antagonist. Part 2. Identification and optimization of triazolobenzazepines. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Vasopressin V1a receptor | |||
| Name: | Vasopressin V1a receptor | ||
| Synonyms: | AVPR V1a | AVPR1 | AVPR1A | Antidiuretic hormone receptor 1a | V1AR_HUMAN | V1aR | VASOPRESSIN V1A | Vascular/hepatic-type arginine vasopressin receptor | Vasopressin V1 receptor | Vasopressin V1a receptor | Vasopressin receptor | ||
| Type: | Receptor | ||
| Mol. Mass.: | 46820.18 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P37288 | ||
| Residue: | 418 | ||
| Sequence: |
| ||
| BDBM50546419 | |||
| n/a | |||
| Name | BDBM50546419 | ||
| Synonyms: | CHEMBL4790066 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C21H21ClN4O | ||
| Mol. Mass. | 380.871 | ||
| SMILES | Cc1cc(no1)[C@H]1CC[C@@H](CC1)c1nnc2C3CC3c3cc(Cl)ccc3-n12 |r,wU:9.13,wD:6.6,(47.62,-42.58,;48.52,-43.82,;48.03,-45.28,;49.28,-46.19,;50.52,-45.29,;50.05,-43.83,;49.27,-47.73,;47.92,-48.5,;47.91,-50.04,;49.25,-50.82,;50.6,-50.06,;50.61,-48.51,;49.26,-52.38,;50.5,-53.28,;50.03,-54.75,;48.49,-54.75,;47.46,-55.9,;46.44,-57.05,;45.95,-55.59,;45.48,-54.12,;43.99,-53.81,;43.49,-52.36,;41.98,-52.06,;44.51,-51.21,;46.02,-51.52,;46.5,-52.97,;48,-53.28,)| | ||
| Structure | 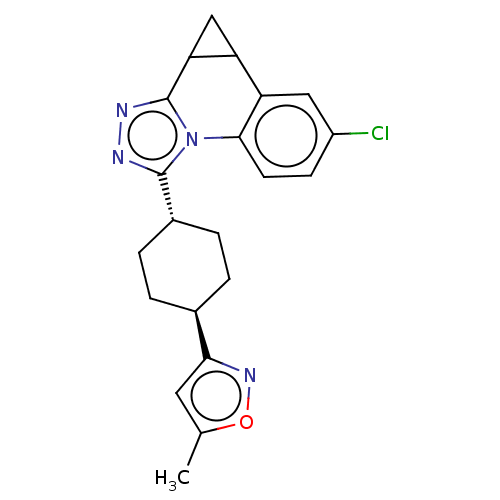
| ||