| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50109453 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_31508 (CHEMBL647493) |
|---|
| Ki | 423.0±n/a nM |
|---|
| Citation |  Baraldi, PG; Cacciari, B; Moro, S; Spalluto, G; Pastorin, G; Da Ros, T; Klotz, KN; Varani, K; Gessi, S; Borea, PA Synthesis, biological activity, and molecular modeling investigation of new pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine derivatives as human A(3) adenosine receptor antagonists. J Med Chem45:770-80 (2002) [PubMed] Baraldi, PG; Cacciari, B; Moro, S; Spalluto, G; Pastorin, G; Da Ros, T; Klotz, KN; Varani, K; Gessi, S; Borea, PA Synthesis, biological activity, and molecular modeling investigation of new pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine derivatives as human A(3) adenosine receptor antagonists. J Med Chem45:770-80 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM50109453 |
|---|
| n/a |
|---|
| Name | BDBM50109453 |
|---|
| Synonyms: | CHEMBL341376 | N-(2-(furan-2-yl)-8-methyl-8H-pyrazolo[4,3-e][1,2,4]triazolo[1,5-c]pyrimidin-5-yl)-2-phenylacetamide | N-(2-Furan-2-yl-8-methyl-8H-pyrazolo[4,3-e][1,2,4]triazolo[1,5-c]pyrimidin-5-yl)-2-phenyl-acetamide | N-[2-Furan-2-yl-8-(3-phenyl-propyl)-2,8-dihydro-3H-pyrazolo[4,3-e][1,2,4]triazolo[1,5-c]pyrimidin-5-yl]-2-phenoxy-acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H15N7O2 |
|---|
| Mol. Mass. | 373.3681 |
|---|
| SMILES | Cn1cc2c(n1)nc(NC(=O)Cc1ccccc1)n1nc(nc21)-c1ccco1 |
|---|
| Structure | 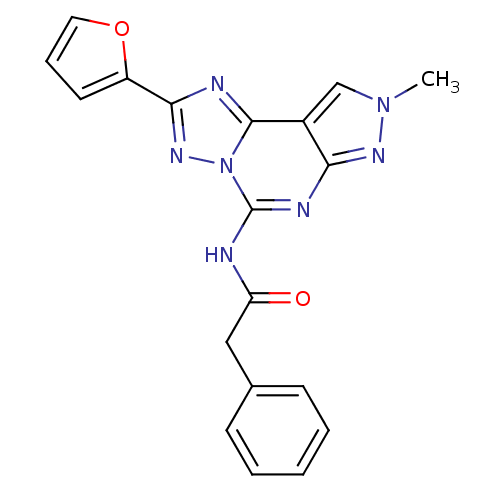
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Baraldi, PG; Cacciari, B; Moro, S; Spalluto, G; Pastorin, G; Da Ros, T; Klotz, KN; Varani, K; Gessi, S; Borea, PA Synthesis, biological activity, and molecular modeling investigation of new pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine derivatives as human A(3) adenosine receptor antagonists. J Med Chem45:770-80 (2002) [PubMed]
Baraldi, PG; Cacciari, B; Moro, S; Spalluto, G; Pastorin, G; Da Ros, T; Klotz, KN; Varani, K; Gessi, S; Borea, PA Synthesis, biological activity, and molecular modeling investigation of new pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine derivatives as human A(3) adenosine receptor antagonists. J Med Chem45:770-80 (2002) [PubMed]