| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-mannosidase |
|---|
| Ligand | BDBM24783 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2029543 (CHEMBL4683701) |
|---|
| IC50 | 49000±n/a nM |
|---|
| Citation |  Luthra, T; Banothu, V; Adepally, U; Kumar, K; M, S; Chakrabarti, S; Maddi, SR; Sen, S Discovery of novel pyrido-pyrrolidine hybrid compounds as alpha-glucosidase inhibitors and alternative agent for control of type 1 diabetes. Eur J Med Chem188:0 (2020) [PubMed] Article Luthra, T; Banothu, V; Adepally, U; Kumar, K; M, S; Chakrabarti, S; Maddi, SR; Sen, S Discovery of novel pyrido-pyrrolidine hybrid compounds as alpha-glucosidase inhibitors and alternative agent for control of type 1 diabetes. Eur J Med Chem188:0 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-mannosidase |
|---|
| Name: | Alpha-mannosidase |
|---|
| Synonyms: | Alpha-mannosidase | Alpha-mannosidase, heavy subunit | Alpha-mannosidase, light subunit | JBM | Jbalpha-man | MANA_CANEN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 108565.93 |
|---|
| Organism: | Canavalia ensiformis |
|---|
| Description: | ChEMBL_109557 |
|---|
| Residue: | 959 |
|---|
| Sequence: | MKYNTGAGTVPEQLNVHLVPHSHDDVGWLKTVDQYYVGSENYIQEACVENVLDSVVMSLQ
RDPNRKFVFGEMAFFHRWWLEQTPETKELKLVKAGQLEFVNGGWCMHDEATTHYIDMIDH
TTLGHRFLQEQFNKIPRAGWQIDPFGHSAVQGYLLGAELGFDSVHFARIDYQDREKRKGE
KSLEVVWRGSKTFGSSAQIFANAFPGHYGPPNGFNFEVRNNFVPLQDDPRLFDTNVEERV
QNFLDAALTQAKLTRTNHLMWTMGDDFQYQYAESWFKQMDKLLHHVNKDGRVNALYSTPS
LYTEAKNAANQTWPLKIDDYFPYADGRNAYWTGFYTSRMLSGYYLATRHSGFFAGKKSTK
YHAFDLADALGIAQHHDAVSGTAKQHTTNDYAKRLALGASKAEAVVSSSLACLTSKQSAD
QCSAPASAFSQCHLFNISYCPPTESSLPDDKSLVVVVYNPLGWSRNEIVRIPVNDANLVV
KDSSGNKLEVQYVEMDDVTANLRSFYVKYWSLFKASVPPLGWSTYFISEATGKGTRNALT
LSQKGETLNIGPGDLKMSFSSLTGQLKRMYNSKTGVDLPIQQNYLWYESSEGDFSDYQAS
GAYLFRPNGQPPPHTVSRSSVTRVTRGPLVDEVHQKFNSWISQVTRLYKDKDHAEIEFTI
GPIPTDDGVGKEVITRMTSTMATNKEFYTDSNGRDFLKRVRDYREDWPLEVTQPVAGNYY
PLNLGLYTKDEKSEFSVLVDRATGGASIKDGEVELMLHRRTLRDDGRGVGEPLDEQVCMN
KEYTCEGLTVRGNYYLSIHKPAGGSRWRRTTGQEIYSPMLLAFTQENMENWKSSHSTKAY
AMDPNYSLPPSVALITLEELDDGLVLLRLAHLYEPSEDAEYSTLTKVELKKLFATQKLEE
LREVSLSANQEKSEMKKMKWSVEGDNEQEPQAVRGGPVSNADFVVELGPMEIRTFLLQF
|
|
|
|---|
| BDBM24783 |
|---|
| n/a |
|---|
| Name | BDBM24783 |
|---|
| Synonyms: | 4H-chromen-4-one | chromone, 12 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C9H6O2 |
|---|
| Mol. Mass. | 146.1427 |
|---|
| SMILES | O=c1ccoc2ccccc12 |
|---|
| Structure | 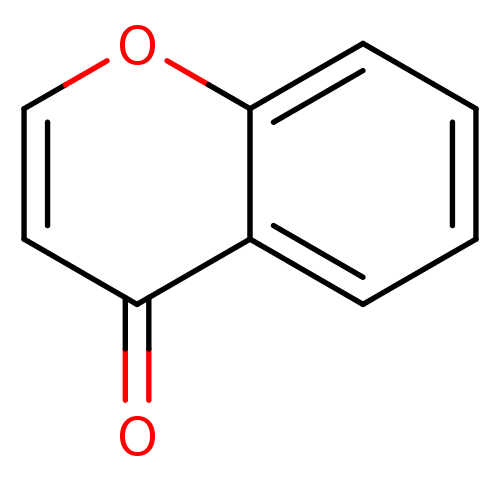
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Luthra, T; Banothu, V; Adepally, U; Kumar, K; M, S; Chakrabarti, S; Maddi, SR; Sen, S Discovery of novel pyrido-pyrrolidine hybrid compounds as alpha-glucosidase inhibitors and alternative agent for control of type 1 diabetes. Eur J Med Chem188:0 (2020) [PubMed] Article
Luthra, T; Banothu, V; Adepally, U; Kumar, K; M, S; Chakrabarti, S; Maddi, SR; Sen, S Discovery of novel pyrido-pyrrolidine hybrid compounds as alpha-glucosidase inhibitors and alternative agent for control of type 1 diabetes. Eur J Med Chem188:0 (2020) [PubMed] Article