| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Monoglyceride lipase |
|---|
| Ligand | BDBM50562161 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2077554 (CHEMBL4733345) |
|---|
| IC50 | 36±n/a nM |
|---|
| Citation |  Granchi, C; Bononi, G; Ferrisi, R; Gori, E; Mantini, G; Glasmacher, S; Poli, G; Palazzolo, S; Caligiuri, I; Rizzolio, F; Canzonieri, V; Perin, T; Gertsch, J; Sodi, A; Giovannetti, E; Macchia, M; Minutolo, F; Tuccinardi, T; Chicca, A Design, synthesis and biological evaluation of second-generation benzoylpiperidine derivatives as reversible monoacylglycerol lipase (MAGL) inhibitors. Eur J Med Chem209:0 (2021) [PubMed] Article Granchi, C; Bononi, G; Ferrisi, R; Gori, E; Mantini, G; Glasmacher, S; Poli, G; Palazzolo, S; Caligiuri, I; Rizzolio, F; Canzonieri, V; Perin, T; Gertsch, J; Sodi, A; Giovannetti, E; Macchia, M; Minutolo, F; Tuccinardi, T; Chicca, A Design, synthesis and biological evaluation of second-generation benzoylpiperidine derivatives as reversible monoacylglycerol lipase (MAGL) inhibitors. Eur J Med Chem209:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Monoglyceride lipase |
|---|
| Name: | Monoglyceride lipase |
|---|
| Synonyms: | HU-K5 | Lysophospholipase homolog | Lysophospholipase-like | MAGL | MGL | MGLL | MGLL_HUMAN |
|---|
| Type: | Hydrolase |
|---|
| Mol. Mass.: | 33264.56 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human recombinant MGL (Cayman Chemical, cat# 10008354). |
|---|
| Residue: | 303 |
|---|
| Sequence: | MPEESSPRRTPQSIPYQDLPHLVNADGQYLFCRYWKPTGTPKALIFVSHGAGEHSGRYEE
LARMLMGLDLLVFAHDHVGHGQSEGERMVVSDFHVFVRDVLQHVDSMQKDYPGLPVFLLG
HSMGGAIAILTAAERPGHFAGMVLISPLVLANPESATTFKVLAAKVLNLVLPNLSLGPID
SSVLSRNKTEVDIYNSDPLICRAGLKVCFGIQLLNAVSRVERALPKLTVPFLLLQGSADR
LCDSKGAYLLMELAKSQDKTLKIYEGAYHVLHKELPEVTNSVFHEINMWVSQRTATAGTA
SPP
|
|
|
|---|
| BDBM50562161 |
|---|
| n/a |
|---|
| Name | BDBM50562161 |
|---|
| Synonyms: | CHEMBL4755606 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H23F2NO3 |
|---|
| Mol. Mass. | 435.4625 |
|---|
| SMILES | Oc1cc(C(=O)N2CCC(CC2)C(=O)c2ccc(Cc3ccccc3)cc2)c(F)cc1F |
|---|
| Structure | 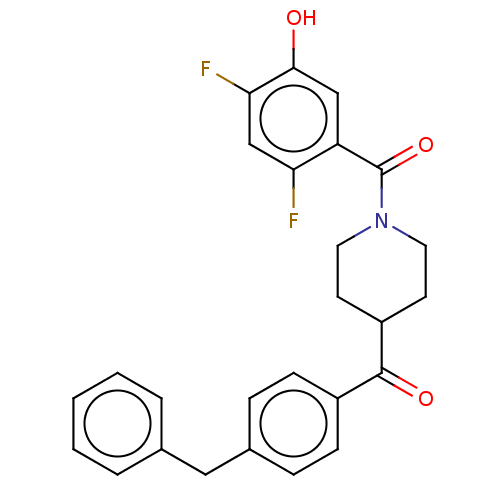
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Granchi, C; Bononi, G; Ferrisi, R; Gori, E; Mantini, G; Glasmacher, S; Poli, G; Palazzolo, S; Caligiuri, I; Rizzolio, F; Canzonieri, V; Perin, T; Gertsch, J; Sodi, A; Giovannetti, E; Macchia, M; Minutolo, F; Tuccinardi, T; Chicca, A Design, synthesis and biological evaluation of second-generation benzoylpiperidine derivatives as reversible monoacylglycerol lipase (MAGL) inhibitors. Eur J Med Chem209:0 (2021) [PubMed] Article
Granchi, C; Bononi, G; Ferrisi, R; Gori, E; Mantini, G; Glasmacher, S; Poli, G; Palazzolo, S; Caligiuri, I; Rizzolio, F; Canzonieri, V; Perin, T; Gertsch, J; Sodi, A; Giovannetti, E; Macchia, M; Minutolo, F; Tuccinardi, T; Chicca, A Design, synthesis and biological evaluation of second-generation benzoylpiperidine derivatives as reversible monoacylglycerol lipase (MAGL) inhibitors. Eur J Med Chem209:0 (2021) [PubMed] Article