| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sodium- and chloride-dependent GABA transporter 1 |
|---|
| Ligand | BDBM50577075 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2129151 (CHEMBL4838580) |
|---|
| IC50 | 10000±n/a nM |
|---|
| Citation |  Zar?ba, P; Sa?at, K; H�fner, GC; ??tka, K; Bajda, M; Latacz, G; Kotniewicz, K; Rapacz, A; Podkowa, A; Maj, M; J�?wiak, K; Filipek, B; Wanner, KT; Malawska, B; Kulig, K Development of tricyclic N-benzyl-4-hydroxybutanamide derivatives as inhibitors of GABA transporters mGAT1-4 with anticonvulsant, antinociceptive, and antidepressant activity. Eur J Med Chem221:0 (2021) [PubMed] Article Zar?ba, P; Sa?at, K; H�fner, GC; ??tka, K; Bajda, M; Latacz, G; Kotniewicz, K; Rapacz, A; Podkowa, A; Maj, M; J�?wiak, K; Filipek, B; Wanner, KT; Malawska, B; Kulig, K Development of tricyclic N-benzyl-4-hydroxybutanamide derivatives as inhibitors of GABA transporters mGAT1-4 with anticonvulsant, antinociceptive, and antidepressant activity. Eur J Med Chem221:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sodium- and chloride-dependent GABA transporter 1 |
|---|
| Name: | Sodium- and chloride-dependent GABA transporter 1 |
|---|
| Synonyms: | GABA transporter | Gabt1 | Gat-1 | Gat1 | SC6A1_RAT | Slc6a1 | Solute carrier family 6 member 1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 67006.26 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1472611 |
|---|
| Residue: | 599 |
|---|
| Sequence: | MATDNSKVADGQISTEVSEAPVASDKPKTLVVKVQKKAGDLPDRDTWKGRFDFLMSCVGY
AIGLGNVWRFPYLCGKNGGGAFLIPYFLTLIFAGVPLFLLECSLGQYTSIGGLGVWKLAP
MFKGVGLAAAVLSFWLNIYYIVIISWAIYYLYNSFTTTLPWKQCDNPWNTDRCFSNYSLV
NTTNMTSAVVEFWERNMHQMTDGLDKPGQIRWPLAITLAIAWVLVYFCIWKGVGWTGKVV
YFSATYPYIMLIILFFRGVTLPGAKEGILFYITPNFRKLSDSEVWLDAATQIFFSYGLGL
GSLIALGSYNSFHNNVYRDSIIVCCINSCTSMFAGFVIFSIVGFMAHVTKRSIADVAASG
PGLAFLAYPEAVTQLPISPLWAILFFSMLLMLGIDSQFCTVEGFITALVDEYPRLLRNRR
ELFIAAVCIVSYLIGLSNITQGGIYVFKLFDYYSASGMSLLFLVFFECVSISWFYGVNRF
YDNIQEMVGSRPCIWWKLCWSFFTPIIVAGVFLFSAVQMTPLTMGSYVFPKWGQGVGWLM
ALSSMVLIPGYMAYMFLTLKGSLKQRLQVMIQPSEDIVRPENGPEQPQAGSSASKEAYI
|
|
|
|---|
| BDBM50577075 |
|---|
| n/a |
|---|
| Name | BDBM50577075 |
|---|
| Synonyms: | CHEMBL4856428 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H33ClN2O2 |
|---|
| Mol. Mass. | 489.048 |
|---|
| SMILES | [#6]-[#7](-[#6]-[#6]\[#6]=[#6]-1/c2ccccc2-[#6]-[#6]-c2ccccc-12)-[#6](-[#6]-[#6]-[#8])-[#6](=O)-[#7]-[#6]-c1ccc(Cl)cc1 |
|---|
| Structure | 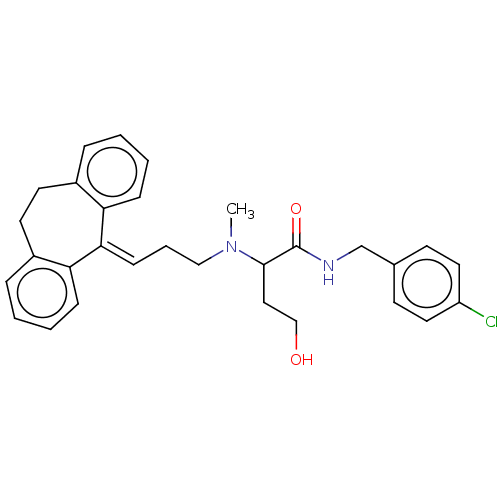
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zar?ba, P; Sa?at, K; H�fner, GC; ??tka, K; Bajda, M; Latacz, G; Kotniewicz, K; Rapacz, A; Podkowa, A; Maj, M; J�?wiak, K; Filipek, B; Wanner, KT; Malawska, B; Kulig, K Development of tricyclic N-benzyl-4-hydroxybutanamide derivatives as inhibitors of GABA transporters mGAT1-4 with anticonvulsant, antinociceptive, and antidepressant activity. Eur J Med Chem221:0 (2021) [PubMed] Article
Zar?ba, P; Sa?at, K; H�fner, GC; ??tka, K; Bajda, M; Latacz, G; Kotniewicz, K; Rapacz, A; Podkowa, A; Maj, M; J�?wiak, K; Filipek, B; Wanner, KT; Malawska, B; Kulig, K Development of tricyclic N-benzyl-4-hydroxybutanamide derivatives as inhibitors of GABA transporters mGAT1-4 with anticonvulsant, antinociceptive, and antidepressant activity. Eur J Med Chem221:0 (2021) [PubMed] Article