| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Ligand | BDBM4552 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2218428 (CHEMBL5131762) |
|---|
| IC50 | 1.2±n/a nM |
|---|
| Citation |  Raghuvanshi, R; Bharate, SB Recent Developments in the Use of Kinase Inhibitors for Management of Viral Infections. J Med Chem65:893-921 (2022) [PubMed] Article Raghuvanshi, R; Bharate, SB Recent Developments in the Use of Kinase Inhibitors for Management of Viral Infections. J Med Chem65:893-921 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Proto-oncogene tyrosine-protein kinase Src |
|---|
| Name: | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Synonyms: | Calmodulin/Proto-oncogene tyrosine-protein kinase Src | Protein cereblon/Tyrosine-protein kinase SRC | Proto-oncogene c-Src | Proto-oncogene tyrosine-protein kinase Src (c-Src) | SRC | SRC1 | SRC_HUMAN | Tyrosine-protein kinase Src (SRC) | V-src sarcoma (Schmidt-Ruppin A-2) viral oncogene homolog (avian) | c-Src | p60-Src | pp60c-src |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 59838.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P12931 |
|---|
| Residue: | 536 |
|---|
| Sequence: | MGSNKSKPKDASQRRRSLEPAENVHGAGGGAFPASQTPSKPASADGHRGPSAAFAPAAAE
PKLFGGFNSSDTVTSPQRAGPLAGGVTTFVALYDYESRTETDLSFKKGERLQIVNNTEGD
WWLAHSLSTGQTGYIPSNYVAPSDSIQAEEWYFGKITRRESERLLLNAENPRGTFLVRES
ETTKGAYCLSVSDFDNAKGLNVKHYKIRKLDSGGFYITSRTQFNSLQQLVAYYSKHADGL
CHRLTTVCPTSKPQTQGLAKDAWEIPRESLRLEVKLGQGCFGEVWMGTWNGTTRVAIKTL
KPGTMSPEAFLQEAQVMKKLRHEKLVQLYAVVSEEPIYIVTEYMSKGSLLDFLKGETGKY
LRLPQLVDMAAQIASGMAYVERMNYVHRDLRAANILVGENLVCKVADFGLARLIEDNEYT
ARQGAKFPIKWTAPEAALYGRFTIKSDVWSFGILLTELTTKGRVPYPGMVNREVLDQVER
GYRMPCPPECPESLHDLMCQCWRKEPEERPTFEYLQAFLEDYFTSTEPQYQPGENL
|
|
|
|---|
| BDBM4552 |
|---|
| n/a |
|---|
| Name | BDBM4552 |
|---|
| Synonyms: | 4-[(2,4-Dichloro-5-methoxyphenyl)amino]-6-methoxy-7-[3-(4-methyl-1-piperazinyl)propoxy]-3-quinolinecarbonitrile | 4-[(2,4-dichloro-5-methoxyphenyl)amino]-6-methoxy-7-[3-(4-methylpiperazin-1-yl)propoxy]quinoline-3-carbonitrile | BOSUTINIB | CHEMBL288441 | SKI-606 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H29Cl2N5O3 |
|---|
| Mol. Mass. | 530.446 |
|---|
| SMILES | COc1cc(Nc2c(cnc3cc(OCCCN4CCN(C)CC4)c(OC)cc23)C#N)c(Cl)cc1Cl |
|---|
| Structure | 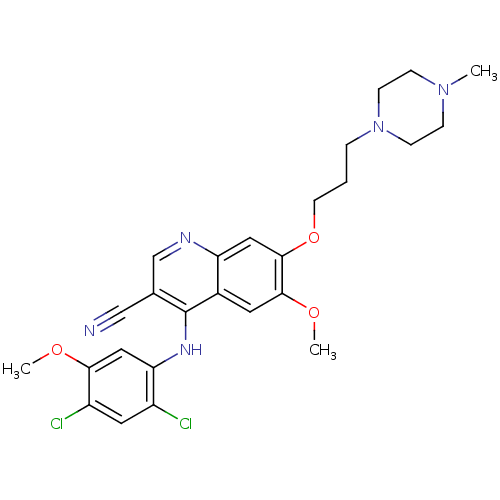
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Raghuvanshi, R; Bharate, SB Recent Developments in the Use of Kinase Inhibitors for Management of Viral Infections. J Med Chem65:893-921 (2022) [PubMed] Article
Raghuvanshi, R; Bharate, SB Recent Developments in the Use of Kinase Inhibitors for Management of Viral Infections. J Med Chem65:893-921 (2022) [PubMed] Article