| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Somatostatin receptor type 1 |
|---|
| Ligand | BDBM523767 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2223693 (CHEMBL5137206) |
|---|
| Ki | 468±n/a nM |
|---|
| Citation |  Neumann, WL; Sandoval, KE; Mobayen, S; Minaeian, M; Kukielski, SG; Srabony, KN; Frare, R; Slater, O; Farr, SA; Niehoff, ML; Hospital, A; Kontoyianni, M; Crider, AM; Witt, KA Synthesis and structure-activity relationships of 3,4,5-trisubstituted-1,2,4-triazoles: high affinity and selective somatostatin receptor-4 agonists for Alzheimer's disease treatment. RSC Med Chem12:1352-1365 (2021) [PubMed] Article Neumann, WL; Sandoval, KE; Mobayen, S; Minaeian, M; Kukielski, SG; Srabony, KN; Frare, R; Slater, O; Farr, SA; Niehoff, ML; Hospital, A; Kontoyianni, M; Crider, AM; Witt, KA Synthesis and structure-activity relationships of 3,4,5-trisubstituted-1,2,4-triazoles: high affinity and selective somatostatin receptor-4 agonists for Alzheimer's disease treatment. RSC Med Chem12:1352-1365 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Somatostatin receptor type 1 |
|---|
| Name: | Somatostatin receptor type 1 |
|---|
| Synonyms: | SOMATOSTATIN SST1 | SRIF-2 | SS-1-R | SS1-R | SS1R | SSR1_HUMAN | SSTR1 | Somatostatin receptor type 1 (SSTR1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 42692.81 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P30872 |
|---|
| Residue: | 391 |
|---|
| Sequence: | MFPNGTASSPSSSPSPSPGSCGEGGGSRGPGAGAADGMEEPGRNASQNGTLSEGQGSAIL
ISFIYSVVCLVGLCGNSMVIYVILRYAKMKTATNIYILNLAIADELLMLSVPFLVTSTLL
RHWPFGALLCRLVLSVDAVNMFTSIYCLTVLSVDRYVAVVHPIKAARYRRPTVAKVVNLG
VWVLSLLVILPIVVFSRTAANSDGTVACNMLMPEPAQRWLVGFVLYTFLMGFLLPVGAIC
LCYVLIIAKMRMVALKAGWQQRKRSERKITLMVMMVVMVFVICWMPFYVVQLVNVFAEQD
DATVSQLSVILGYANSCANPILYGFLSDNFKRSFQRILCLSWMDNAAEEPVDYYATALKS
RAYSVEDFQPENLESGGVFRNGTCTSRITTL
|
|
|
|---|
| BDBM523767 |
|---|
| n/a |
|---|
| Name | BDBM523767 |
|---|
| Synonyms: | US11136312, Compound SK-I-91 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H22F2N6 |
|---|
| Mol. Mass. | 432.4685 |
|---|
| SMILES | Fc1ccc(Cc2nnc(Cc3c[nH]c4cc(F)ccc34)n2CCCc2c[nH]cn2)cc1 |
|---|
| Structure | 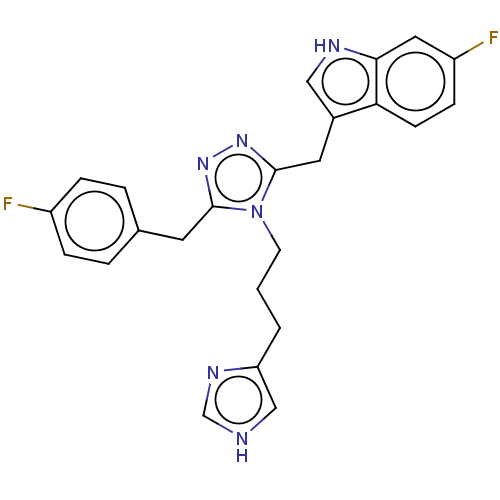
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Neumann, WL; Sandoval, KE; Mobayen, S; Minaeian, M; Kukielski, SG; Srabony, KN; Frare, R; Slater, O; Farr, SA; Niehoff, ML; Hospital, A; Kontoyianni, M; Crider, AM; Witt, KA Synthesis and structure-activity relationships of 3,4,5-trisubstituted-1,2,4-triazoles: high affinity and selective somatostatin receptor-4 agonists for Alzheimer's disease treatment. RSC Med Chem12:1352-1365 (2021) [PubMed] Article
Neumann, WL; Sandoval, KE; Mobayen, S; Minaeian, M; Kukielski, SG; Srabony, KN; Frare, R; Slater, O; Farr, SA; Niehoff, ML; Hospital, A; Kontoyianni, M; Crider, AM; Witt, KA Synthesis and structure-activity relationships of 3,4,5-trisubstituted-1,2,4-triazoles: high affinity and selective somatostatin receptor-4 agonists for Alzheimer's disease treatment. RSC Med Chem12:1352-1365 (2021) [PubMed] Article