| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50420191 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2226863 (CHEMBL5140376) |
|---|
| IC50 | 4.9±n/a nM |
|---|
| Citation |  Ling, X; Hao, QQ; Pannecouque, C; Clercq, E; Chen, FE Expansion of the S-CN-DABO scaffold to exploit the impact on inhibitory activities against the non-nucleoside HIV-1 reverse transcriptase. Eur J Med Chem238:0 (2022) [PubMed] Article Ling, X; Hao, QQ; Pannecouque, C; Clercq, E; Chen, FE Expansion of the S-CN-DABO scaffold to exploit the impact on inhibitory activities against the non-nucleoside HIV-1 reverse transcriptase. Eur J Med Chem238:0 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50420191 |
|---|
| n/a |
|---|
| Name | BDBM50420191 |
|---|
| Synonyms: | ACETPHENETIDIN | Acetophenetidin | PHENACETIN |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H13NO2 |
|---|
| Mol. Mass. | 179.2157 |
|---|
| SMILES | CCOc1ccc(NC(C)=O)cc1 |
|---|
| Structure | 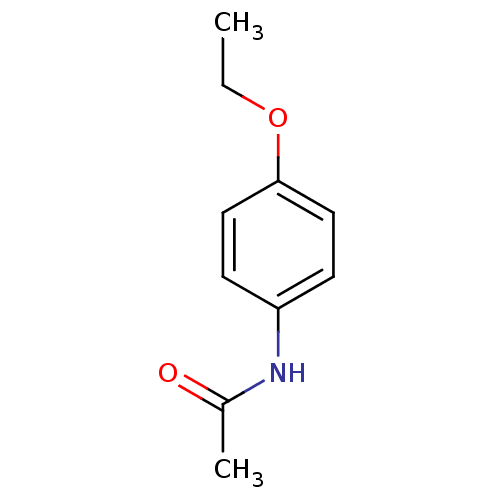
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ling, X; Hao, QQ; Pannecouque, C; Clercq, E; Chen, FE Expansion of the S-CN-DABO scaffold to exploit the impact on inhibitory activities against the non-nucleoside HIV-1 reverse transcriptase. Eur J Med Chem238:0 (2022) [PubMed] Article
Ling, X; Hao, QQ; Pannecouque, C; Clercq, E; Chen, FE Expansion of the S-CN-DABO scaffold to exploit the impact on inhibitory activities against the non-nucleoside HIV-1 reverse transcriptase. Eur J Med Chem238:0 (2022) [PubMed] Article