| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lysine-specific histone demethylase 2 |
|---|
| Ligand | BDBM50603922 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2245503 (CHEMBL5159713) |
|---|
| IC50 | 1010±n/a nM |
|---|
| Citation |  Li, C; Su, M; Zhu, W; Kan, W; Ge, T; Xu, G; Wang, S; Sheng, L; Gao, F; Ye, Y; Wang, J; Zhou, Y; Li, J; Liu, H Structure-Activity Relationship Study of Indolin-5-yl-cyclopropanamine Derivatives as Selective Lysine Specific Demethylase 1 (LSD1) Inhibitors. J Med Chem65:4335-4349 (2022) [PubMed] Article Li, C; Su, M; Zhu, W; Kan, W; Ge, T; Xu, G; Wang, S; Sheng, L; Gao, F; Ye, Y; Wang, J; Zhou, Y; Li, J; Liu, H Structure-Activity Relationship Study of Indolin-5-yl-cyclopropanamine Derivatives as Selective Lysine Specific Demethylase 1 (LSD1) Inhibitors. J Med Chem65:4335-4349 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lysine-specific histone demethylase 2 |
|---|
| Name: | Lysine-specific histone demethylase 2 |
|---|
| Synonyms: | AOF1 | C6orf193 | Flavin-containing amine oxidase domain-containing protein 1 | KDM1B | KDM1B_HUMAN | LSD2 | Lysine-specific histone demethylase 1B | Lysine-specific histone demethylase 2 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 92112.91 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_992590 |
|---|
| Residue: | 822 |
|---|
| Sequence: | MATPRGRTKKKASFDHSPDSLPLRSSGRQAKKKATETTDEDEDGGSEKKYRKCEKAGCTA
TCPVCFASASERCAKNGYTSRWYHLSCGEHFCNECFDHYYRSHKDGYDKYTTWKKIWTSN
GKTEPSPKAFMADQQLPYWVQCTKPECRKWRQLTKEIQLTPQIAKTYRCGMKPNTAIKPE
TSDHCSLPEDLRVLEVSNHWWYSMLILPPLLKDSVAAPLLSAYYPDCVGMSPSCTSTNRA
AATGNASPGKLEHSKAALSVHVPGMNRYFQPFYQPNECGKALCVRPDVMELDELYEFPEY
SRDPTMYLALRNLILALWYTNCKEALTPQKCIPHIIVRGLVRIRCVQEVERILYFMTRKG
LINTGVLSVGADQYLLPKDYHNKSVIIIGAGPAGLAAARQLHNFGIKVTVLEAKDRIGGR
VWDDKSFKGVTVGRGAQIVNGCINNPVALMCEQLGISMHKFGERCDLIQEGGRITDPTID
KRMDFHFNALLDVVSEWRKDKTQLQDVPLGEKIEEIYKAFIKESGIQFSELEGQVLQFHL
SNLEYACGSNLHQVSARSWDHNEFFAQFAGDHTLLTPGYSVIIEKLAEGLDIQLKSPVQC
IDYSGDEVQVTTTDGTGYSAQKVLVTVPLALLQKGAIQFNPPLSEKKMKAINSLGAGIIE
KIALQFPYRFWDSKVQGADFFGHVPPSASKRGLFAVFYDMDPQKKHSVLMSVIAGEAVAS
VRTLDDKQVLQQCMATLRELFKEQEVPDPTKYFVTRWSTDPWIQMAYSFVKTGGSGEAYD
IIAEDIQGTVFFAGEATNRHFPQTVTGAYLSGVREASKIAAF
|
|
|
|---|
| BDBM50603922 |
|---|
| n/a |
|---|
| Name | BDBM50603922 |
|---|
| Synonyms: | CHEMBL5181753 | US11873292, Compound A38 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H28ClN3O2S |
|---|
| Mol. Mass. | 446.005 |
|---|
| SMILES | Clc1cccc(c1)S(=O)(=O)N1CCc2cc(ccc12)C1CC1NCC1CCNCC1 |
|---|
| Structure | 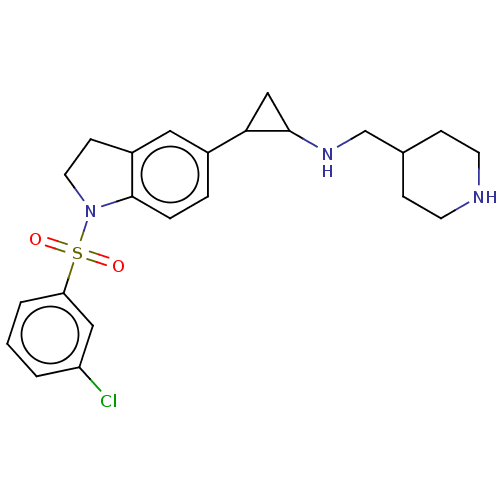
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, C; Su, M; Zhu, W; Kan, W; Ge, T; Xu, G; Wang, S; Sheng, L; Gao, F; Ye, Y; Wang, J; Zhou, Y; Li, J; Liu, H Structure-Activity Relationship Study of Indolin-5-yl-cyclopropanamine Derivatives as Selective Lysine Specific Demethylase 1 (LSD1) Inhibitors. J Med Chem65:4335-4349 (2022) [PubMed] Article
Li, C; Su, M; Zhu, W; Kan, W; Ge, T; Xu, G; Wang, S; Sheng, L; Gao, F; Ye, Y; Wang, J; Zhou, Y; Li, J; Liu, H Structure-Activity Relationship Study of Indolin-5-yl-cyclopropanamine Derivatives as Selective Lysine Specific Demethylase 1 (LSD1) Inhibitors. J Med Chem65:4335-4349 (2022) [PubMed] Article