| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50185962 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_351942 (CHEMBL870788) |
|---|
| IC50 | 730±n/a nM |
|---|
| Citation |  Imamura, S; Ichikawa, T; Nishikawa, Y; Kanzaki, N; Takashima, K; Niwa, S; Iizawa, Y; Baba, M; Sugihara, Y Discovery of a piperidine-4-carboxamide CCR5 antagonist (TAK-220) with highly potent Anti-HIV-1 activity. J Med Chem49:2784-93 (2006) [PubMed] Article Imamura, S; Ichikawa, T; Nishikawa, Y; Kanzaki, N; Takashima, K; Niwa, S; Iizawa, Y; Baba, M; Sugihara, Y Discovery of a piperidine-4-carboxamide CCR5 antagonist (TAK-220) with highly potent Anti-HIV-1 activity. J Med Chem49:2784-93 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50185962 |
|---|
| n/a |
|---|
| Name | BDBM50185962 |
|---|
| Synonyms: | CHEMBL210327 | N-(3,4-dichlorophenyl)-N-(3-{4-[(4-fluorobenzoyl)amino]-piperidin-1-yl}propyl)-1-(methylsulfonyl)piperidine-4-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H35Cl2FN4O4S |
|---|
| Mol. Mass. | 613.571 |
|---|
| SMILES | CS(=O)(=O)N1CCC(CC1)C(=O)N(CCCN1CCC(CC1)NC(=O)c1ccc(F)cc1)c1ccc(Cl)c(Cl)c1 |
|---|
| Structure | 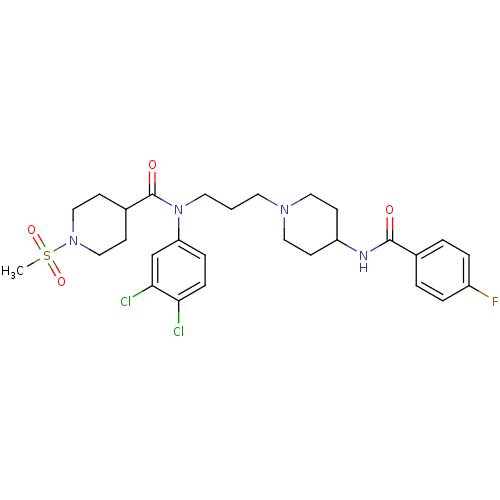
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Imamura, S; Ichikawa, T; Nishikawa, Y; Kanzaki, N; Takashima, K; Niwa, S; Iizawa, Y; Baba, M; Sugihara, Y Discovery of a piperidine-4-carboxamide CCR5 antagonist (TAK-220) with highly potent Anti-HIV-1 activity. J Med Chem49:2784-93 (2006) [PubMed] Article
Imamura, S; Ichikawa, T; Nishikawa, Y; Kanzaki, N; Takashima, K; Niwa, S; Iizawa, Y; Baba, M; Sugihara, Y Discovery of a piperidine-4-carboxamide CCR5 antagonist (TAK-220) with highly potent Anti-HIV-1 activity. J Med Chem49:2784-93 (2006) [PubMed] Article