| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sterol O-acyltransferase 2 |
|---|
| Ligand | BDBM50193718 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_424061 (CHEMBL855070) |
|---|
| IC50 | 41600±n/a nM |
|---|
| Citation |  Park, KH; Park, YD; Han, JM; Im, KR; Lee, BW; Jeong, IY; Jeong, TS; Lee, WS Anti-atherosclerotic and anti-inflammatory activities of catecholic xanthones and flavonoids isolated from Cudrania tricuspidata. Bioorg Med Chem Lett16:5580-3 (2006) [PubMed] Article Park, KH; Park, YD; Han, JM; Im, KR; Lee, BW; Jeong, IY; Jeong, TS; Lee, WS Anti-atherosclerotic and anti-inflammatory activities of catecholic xanthones and flavonoids isolated from Cudrania tricuspidata. Bioorg Med Chem Lett16:5580-3 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sterol O-acyltransferase 2 |
|---|
| Name: | Sterol O-acyltransferase 2 |
|---|
| Synonyms: | ACACT2 | ACAT-2 | ACAT2 | Acetyl-CoA acetyltransferase, cytosolic | Acetyl-CoA transferase-like protein | Acyl coenzyme A:cholesterol acyltransferase 2 | Cholesterol acyltransferase 2 | Cytosolic acetoacetyl-CoA thiolase | SOAT2 | SOAT2_HUMAN |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 59912.75 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O75908 |
|---|
| Residue: | 522 |
|---|
| Sequence: | MEPGGARLRLQRTEGLGGERERQPCGDGNTETHRAPDLVQWTRHMEAVKAQLLEQAQGQL
RELLDRAMREAIQSYPSQDKPLPPPPPGSLSRTQEPSLGKQKVFIIRKSLLDELMEVQHF
RTIYHMFIAGLCVFIISTLAIDFIDEGRLLLEFDLLIFSFGQLPLALVTWVPMFLSTLLA
PYQALRLWARGTWTQATGLGCALLAAHAVVLCALPVHVAVEHQLPPASRCVLVFEQVRFL
MKSYSFLREAVPGTLRARRGEGIQAPSFSSYLYFLFCPTLIYRETYPRTPYVRWNYVAKN
FAQALGCVLYACFILGRLCVPVFANMSREPFSTRALVLSILHATLPGIFMLLLIFFAFLH
CWLNAFAEMLRFGDRMFYRDWWNSTSFSNYYRTWNVVVHDWLYSYVYQDGLRLLGARARG
VAMLGVFLVSAVAHEYIFCFVLGFFYPVMLILFLVIGGMLNFMMHDQRTGPAWNVLMWTM
LFLGQGIQVSLYCQEWYARRHCPLPQATFWGLVTPRSWSCHT
|
|
|
|---|
| BDBM50193718 |
|---|
| n/a |
|---|
| Name | BDBM50193718 |
|---|
| Synonyms: | 2-(2,4-dihydroxy-phenyl)-5-hydroxy-8,8-dimethyl-3-(3-methyl-but-2-enyl)-8H-pyrano[3,2-g]chromen-4-one | 2-(2,4-dihydroxyphenyl)-5-hydroxy-8,8-dimethyl-3-(3-methylbut-2-enyl)pyrano[3,2-g]chromen-4(8H)-one | CHEMBL221907 | cid_5319925 | cudraflavone B |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H24O6 |
|---|
| Mol. Mass. | 420.4545 |
|---|
| SMILES | [#6]\[#6](-[#6])=[#6]\[#6]-c1c(oc2cc3-[#8]C([#6])([#6])[#6]=[#6]-c3c(-[#8])c2c1=O)-c1ccc(-[#8])cc1-[#8] |c:15| |
|---|
| Structure | 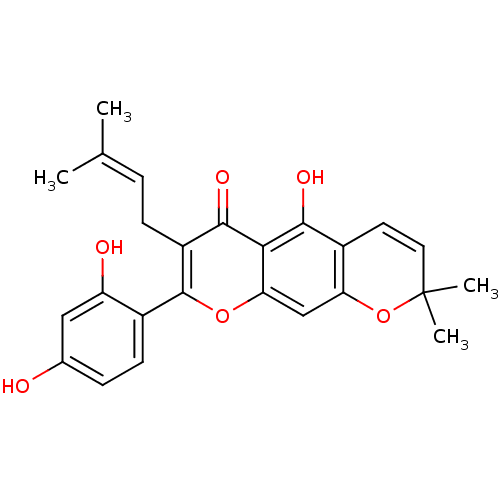
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Park, KH; Park, YD; Han, JM; Im, KR; Lee, BW; Jeong, IY; Jeong, TS; Lee, WS Anti-atherosclerotic and anti-inflammatory activities of catecholic xanthones and flavonoids isolated from Cudrania tricuspidata. Bioorg Med Chem Lett16:5580-3 (2006) [PubMed] Article
Park, KH; Park, YD; Han, JM; Im, KR; Lee, BW; Jeong, IY; Jeong, TS; Lee, WS Anti-atherosclerotic and anti-inflammatory activities of catecholic xanthones and flavonoids isolated from Cudrania tricuspidata. Bioorg Med Chem Lett16:5580-3 (2006) [PubMed] Article