

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Melanocortin receptor 4 | ||
| Ligand | BDBM50217644 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_448693 (CHEMBL897839) | ||
| Ki | 1500±n/a nM | ||
| Citation |  Mutulis, F; Kreicberga, J; Yahorava, S; Mutule, I; Borisova-Jan, L; Yahorau, A; Muceniece, R; Azena, S; Veiksina, S; Petrovska, R; Wikberg, JE Design and synthesis of a library of tertiary amides: evaluation as mimetics of the melanocortins' active core. Bioorg Med Chem15:5787-810 (2007) [PubMed] Article Mutulis, F; Kreicberga, J; Yahorava, S; Mutule, I; Borisova-Jan, L; Yahorau, A; Muceniece, R; Azena, S; Veiksina, S; Petrovska, R; Wikberg, JE Design and synthesis of a library of tertiary amides: evaluation as mimetics of the melanocortins' active core. Bioorg Med Chem15:5787-810 (2007) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Melanocortin receptor 4 | |||
| Name: | Melanocortin receptor 4 | ||
| Synonyms: | MC4-R | MC4R | MC4R_HUMAN | Melanocortin MC4 | Melanocortin receptor 4 (MC-4) | Melanocortin receptor 4 (MC4-R) | Melanocortin receptor 4 (MC4R) | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 36949.50 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P32245 | ||
| Residue: | 332 | ||
| Sequence: |
| ||
| BDBM50217644 | |||
| n/a | |||
| Name | BDBM50217644 | ||
| Synonyms: | CHEMBL397550 | N-(2-chloro-4,5-dimethoxybenzyl)-N-(4-((4-aminocyclohexyl)methyl)cyclohexyl)-4-(1H-indol-3-yl)butanamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C34H46ClN3O3 | ||
| Mol. Mass. | 580.2 | ||
| SMILES | COc1cc(Cl)c(CN(C2CCC(CC3CCC(N)CC3)CC2)C(=O)CCCc2c[nH]c3ccccc23)cc1OC |(20.46,-54,;20.49,-55.54,;21.83,-56.29,;21.86,-57.84,;23.2,-58.58,;23.23,-60.12,;24.52,-57.79,;25.87,-58.54,;27.19,-57.75,;27.16,-56.19,;28.5,-55.4,;28.48,-53.85,;27.13,-53.09,;27.11,-51.55,;28.44,-50.76,;29.78,-51.53,;31.1,-50.75,;31.09,-49.21,;32.42,-48.42,;29.75,-48.45,;28.42,-49.23,;25.79,-53.88,;25.8,-55.44,;28.54,-58.51,;28.57,-60.06,;29.87,-57.71,;31.21,-58.44,;32.55,-57.67,;33.9,-58.4,;35.3,-57.76,;36.34,-58.89,;35.59,-60.24,;36.08,-61.68,;35.08,-62.84,;33.56,-62.54,;33.07,-61.09,;34.08,-59.93,;24.5,-56.25,;23.16,-55.5,;23.14,-53.96,;24.46,-53.17,)| | ||
| Structure | 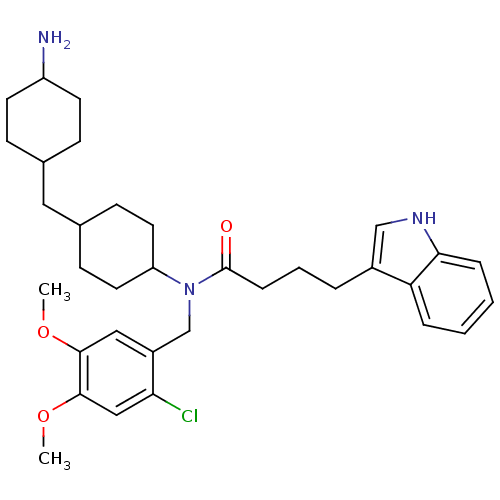
| ||