| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1D |
|---|
| Ligand | BDBM50290916 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1732 |
|---|
| Ki | 3.6±n/a nM |
|---|
| Citation |  Jorand-Lebrun, C; Pauwels, PJ; Palmier, C; Chopin, P; Moret, C; Marien, M; Halazy, S Arylpiperazide derivatives of phenylpiperazines as a new class of potent and selective 5-HT1B receptor antagonists Bioorg Med Chem Lett7:3183-3188 (1997) Article Jorand-Lebrun, C; Pauwels, PJ; Palmier, C; Chopin, P; Moret, C; Marien, M; Halazy, S Arylpiperazide derivatives of phenylpiperazines as a new class of potent and selective 5-HT1B receptor antagonists Bioorg Med Chem Lett7:3183-3188 (1997) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1D |
|---|
| Name: | 5-hydroxytryptamine receptor 1D |
|---|
| Synonyms: | 5-HT-1D | 5-HT-1D-alpha | 5-HT1D | 5-hydroxytryptamine receptor 1D (5-HT1D) | 5HT1D_HUMAN | HTR1D | HTR1DA | HTRL | Serotonin (5-HT) receptor | Serotonin Receptor 1D |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 41920.63 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells. |
|---|
| Residue: | 377 |
|---|
| Sequence: | MSPLNQSAEGLPQEASNRSLNATETSEAWDPRTLQALKISLAVVLSVITLATVLSNAFVL
TTILLTRKLHTPANYLIGSLATTDLLVSILVMPISIAYTITHTWNFGQILCDIWLSSDIT
CCTASILHLCVIALDRYWAITDALEYSKRRTAGHAATMIAIVWAISICISIPPLFWRQAK
AQEEMSDCLVNTSQISYTIYSTCGAFYIPSVLLIILYGRIYRAARNRILNPPSLYGKRFT
TAHLITGSAGSSLCSLNSSLHEGHSHSAGSPLFFNHVKIKLADSALERKRISAARERKAT
KILGIILGAFIICWLPFFVVSLVLPICRDSCWIHPALFDFFTWLGYLNSLINPIIYTVFN
EEFRQAFQKIVPFRKAS
|
|
|
|---|
| BDBM50290916 |
|---|
| n/a |
|---|
| Name | BDBM50290916 |
|---|
| Synonyms: | 4-o-Tolyl-piperazine-1-carboxylic acid 4-chloro-3-(4-methyl-piperazin-1-yl)-phenyl ester; compound with (E)-but-2-enedioic acid | CHEMBL107772 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H29ClN4O2 |
|---|
| Mol. Mass. | 428.955 |
|---|
| SMILES | CN1CCN(CC1)c1cc(OC(=O)N2CCN(CC2)c2ccccc2C)ccc1Cl |
|---|
| Structure | 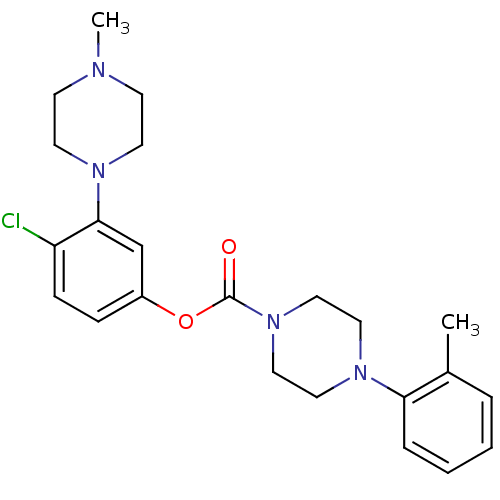
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jorand-Lebrun, C; Pauwels, PJ; Palmier, C; Chopin, P; Moret, C; Marien, M; Halazy, S Arylpiperazide derivatives of phenylpiperazines as a new class of potent and selective 5-HT1B receptor antagonists Bioorg Med Chem Lett7:3183-3188 (1997) Article
Jorand-Lebrun, C; Pauwels, PJ; Palmier, C; Chopin, P; Moret, C; Marien, M; Halazy, S Arylpiperazide derivatives of phenylpiperazines as a new class of potent and selective 5-HT1B receptor antagonists Bioorg Med Chem Lett7:3183-3188 (1997) Article