| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM50300165 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_594165 (CHEMBL1038744) |
|---|
| Ki | 35.8±n/a nM |
|---|
| Citation |  Frecentese, F; Fiorino, F; Perissutti, E; Severino, B; Magli, E; Esposito, A; De Angelis, F; Massarelli, P; Nencini, C; Viti, B; Santagada, V; Caliendo, G Efficient microwave combinatorial synthesis of novel indolic arylpiperazine derivatives as serotoninergic ligands. Eur J Med Chem45:752-9 (2010) [PubMed] Article Frecentese, F; Fiorino, F; Perissutti, E; Severino, B; Magli, E; Esposito, A; De Angelis, F; Massarelli, P; Nencini, C; Viti, B; Santagada, V; Caliendo, G Efficient microwave combinatorial synthesis of novel indolic arylpiperazine derivatives as serotoninergic ligands. Eur J Med Chem45:752-9 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1 | 5-HT1A | 5-Hydroxytryptamine receptor 1A (5-HT1A) | 5-hydroxytryptamine receptor 1A (5HT1A) | 5HT1A_RAT | 5ht1a | G-21 | Htr1a | Serotonin 1 (5-HT1) receptor | Serotonin 1a (5-HT1a) receptor/Adrenergic receptor alpha-1 | Serotonin receptor 1A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46445.29 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Binding assays were performed using rat hippocampal membranes. |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVFSFGQGNNTTASQEPFGTGGNVTSISDVTFSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVRKVEKKGAGT
SLGTSSAPPPKKSLNGQPGSGDWRRCAENRAVGTPCTNGAVRQGDDEATLEVIEVHRVGN
SKEHLPLPSESGSNSYAPACLERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPALLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RR
|
|
|
|---|
| BDBM50300165 |
|---|
| n/a |
|---|
| Name | BDBM50300165 |
|---|
| Synonyms: | CHEMBL566331 | Ethyl 5-(3-(4-(3,4-dichlorophenyl)piperazin-1-yl)propoxy)-1H-indole-2-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H28F3N3O3 |
|---|
| Mol. Mass. | 475.5033 |
|---|
| SMILES | CCOC(=O)c1cc2cc(OCCCN3CCN(CC3)c3cccc(c3)C(F)(F)F)ccc2[nH]1 |
|---|
| Structure | 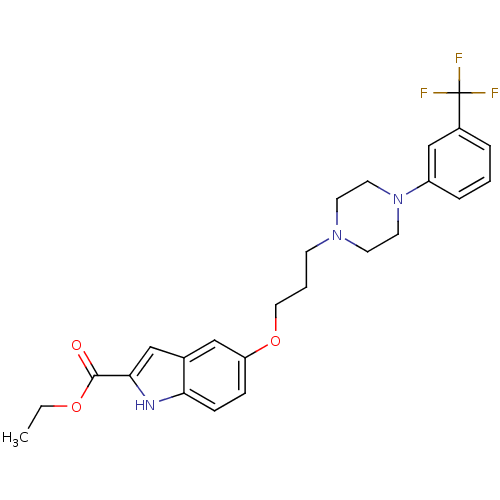
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Frecentese, F; Fiorino, F; Perissutti, E; Severino, B; Magli, E; Esposito, A; De Angelis, F; Massarelli, P; Nencini, C; Viti, B; Santagada, V; Caliendo, G Efficient microwave combinatorial synthesis of novel indolic arylpiperazine derivatives as serotoninergic ligands. Eur J Med Chem45:752-9 (2010) [PubMed] Article
Frecentese, F; Fiorino, F; Perissutti, E; Severino, B; Magli, E; Esposito, A; De Angelis, F; Massarelli, P; Nencini, C; Viti, B; Santagada, V; Caliendo, G Efficient microwave combinatorial synthesis of novel indolic arylpiperazine derivatives as serotoninergic ligands. Eur J Med Chem45:752-9 (2010) [PubMed] Article