| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Amine oxidase [flavin-containing] B |
|---|
| Ligand | BDBM50265779 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_657399 (CHEMBL1246375) |
|---|
| IC50 | 2370±n/a nM |
|---|
| Citation |  Chimenti, F; Secci, D; Bolasco, A; Chimenti, P; Granese, A; Carradori, S; Yáñez, M; Orallo, F; Sanna, ML; Gallinella, B; Cirilli, R Synthesis, stereochemical separation, and biological evaluation of selective inhibitors of human MAO-B: 1-(4-arylthiazol-2-yl)-2-(3-methylcyclohexylidene)hydrazines. J Med Chem53:6516-20 (2010) [PubMed] Article Chimenti, F; Secci, D; Bolasco, A; Chimenti, P; Granese, A; Carradori, S; Yáñez, M; Orallo, F; Sanna, ML; Gallinella, B; Cirilli, R Synthesis, stereochemical separation, and biological evaluation of selective inhibitors of human MAO-B: 1-(4-arylthiazol-2-yl)-2-(3-methylcyclohexylidene)hydrazines. J Med Chem53:6516-20 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Amine oxidase [flavin-containing] B |
|---|
| Name: | Amine oxidase [flavin-containing] B |
|---|
| Synonyms: | AOFB_HUMAN | MAO-B | MAOB | Monoamine oxidase type B | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAOB) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 58768.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P27338 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MSNKCDVVVVGGGISGMAAAKLLHDSGLNVVVLEARDRVGGRTYTLRNQKVKYVDLGGSY
VGPTQNRILRLAKELGLETYKVNEVERLIHHVKGKSYPFRGPFPPVWNPITYLDHNNFWR
TMDDMGREIPSDAPWKAPLAEEWDNMTMKELLDKLCWTESAKQLATLFVNLCVTAETHEV
SALWFLWYVKQCGGTTRIISTTNGGQERKFVGGSGQVSERIMDLLGDRVKLERPVIYIDQ
TRENVLVETLNHEMYEAKYVISAIPPTLGMKIHFNPPLPMMRNQMITRVPLGSVIKCIVY
YKEPFWRKKDYCGTMIIDGEEAPVAYTLDDTKPEGNYAAIMGFILAHKARKLARLTKEER
LKKLCELYAKVLGSLEALEPVHYEEKNWCEEQYSGGCYTTYFPPGILTQYGRVLRQPVDR
IYFAGTETATHWSGYMEGAVEAGERAAREILHAMGKIPEDEIWQSEPESVDVPAQPITTT
FLERHLPSVPGLLRLIGLTTIFSATALGFLAHKRGLLVRV
|
|
|
|---|
| BDBM50265779 |
|---|
| n/a |
|---|
| Name | BDBM50265779 |
|---|
| Synonyms: | (+/-)-1-(4-(4-methoxyphenyl)thiazol-2-yl)-2-(2-methylcyclohexylidene)hydrazine | 4-(4-methoxyphenyl)-2-(2-(2-methylcyclohexylidene)hydrazinyl)thiazole | CHEMBL399353 | CHEMBL457535 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H21N3OS |
|---|
| Mol. Mass. | 315.433 |
|---|
| SMILES | COc1ccc(cc1)-c1csc(NN=C2CCCCC2C)n1 |w:13.13| |
|---|
| Structure | 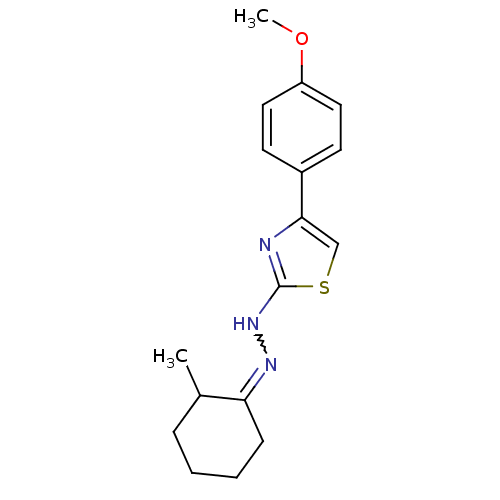
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chimenti, F; Secci, D; Bolasco, A; Chimenti, P; Granese, A; Carradori, S; Yáñez, M; Orallo, F; Sanna, ML; Gallinella, B; Cirilli, R Synthesis, stereochemical separation, and biological evaluation of selective inhibitors of human MAO-B: 1-(4-arylthiazol-2-yl)-2-(3-methylcyclohexylidene)hydrazines. J Med Chem53:6516-20 (2010) [PubMed] Article
Chimenti, F; Secci, D; Bolasco, A; Chimenti, P; Granese, A; Carradori, S; Yáñez, M; Orallo, F; Sanna, ML; Gallinella, B; Cirilli, R Synthesis, stereochemical separation, and biological evaluation of selective inhibitors of human MAO-B: 1-(4-arylthiazol-2-yl)-2-(3-methylcyclohexylidene)hydrazines. J Med Chem53:6516-20 (2010) [PubMed] Article