

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | C-C chemokine receptor type 2 | ||
| Ligand | BDBM50331722 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_687875 (CHEMBL1291439) | ||
| IC50 | 0.950000±n/a nM | ||
| Citation |  Xue, CB; Wang, A; Meloni, D; Zhang, K; Kong, L; Feng, H; Glenn, J; Huang, T; Zhang, Y; Cao, G; Anand, R; Zheng, C; Xia, M; Han, Q; Robinson, DJ; Storace, L; Shao, L; Li, M; Brodmerkel, CM; Covington, M; Scherle, P; Diamond, S; Yeleswaram, S; Vaddi, K; Newton, R; Hollis, G; Friedman, S; Metcalf, B Discovery of INCB3344, a potent, selective and orally bioavailable antagonist of human and murine CCR2. Bioorg Med Chem Lett20:7473-8 (2010) [PubMed] Article Xue, CB; Wang, A; Meloni, D; Zhang, K; Kong, L; Feng, H; Glenn, J; Huang, T; Zhang, Y; Cao, G; Anand, R; Zheng, C; Xia, M; Han, Q; Robinson, DJ; Storace, L; Shao, L; Li, M; Brodmerkel, CM; Covington, M; Scherle, P; Diamond, S; Yeleswaram, S; Vaddi, K; Newton, R; Hollis, G; Friedman, S; Metcalf, B Discovery of INCB3344, a potent, selective and orally bioavailable antagonist of human and murine CCR2. Bioorg Med Chem Lett20:7473-8 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| C-C chemokine receptor type 2 | |||
| Name: | C-C chemokine receptor type 2 | ||
| Synonyms: | C-C chemokine receptor type 2 (CCR2) | CCR2 | CCR2_HUMAN | CMKBR2 | Chemoattractant Cytokine Receptor 2 (CCR2) | Chemokine Receptor Type 2b (CCR2b) | Monocyte chemotactic protein-1 (MCP-1) | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 41932.32 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P41597 | ||
| Residue: | 374 | ||
| Sequence: |
| ||
| BDBM50331722 | |||
| n/a | |||
| Name | BDBM50331722 | ||
| Synonyms: | CHEMBL1290636 | N-(2-((3S,4S)-4-ethoxy-1-(cis-4-phenylcyclohexyl)pyrrolidin-3-ylamino)-2-oxoethyl)-3-(trifluoromethyl)benzamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C28H34F3N3O3 | ||
| Mol. Mass. | 517.5831 | ||
| SMILES | CCO[C@H]1CN(C[C@@H]1NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)[C@H]1CC[C@H](CC1)c1ccccc1 |r,wU:3.2,25.26,28.33,wD:7.8,(24.27,-20.7,;23.02,-19.81,;23.17,-18.27,;21.92,-17.37,;20.46,-17.85,;19.55,-16.6,;20.45,-15.36,;21.91,-15.83,;23.25,-15.06,;24.58,-15.83,;24.58,-17.38,;25.92,-15.06,;27.26,-15.83,;28.59,-15.06,;28.59,-13.52,;29.93,-15.83,;31.25,-15.06,;32.59,-15.83,;32.59,-17.38,;31.25,-18.15,;29.93,-17.38,;31.25,-19.69,;29.93,-20.46,;32.59,-20.46,;31.24,-21.22,;18.01,-16.6,;17.24,-17.94,;15.69,-17.94,;14.93,-16.6,;15.7,-15.27,;17.23,-15.28,;13.39,-16.6,;12.62,-15.26,;11.08,-15.26,;10.31,-16.59,;11.08,-17.93,;12.62,-17.93,)| | ||
| Structure | 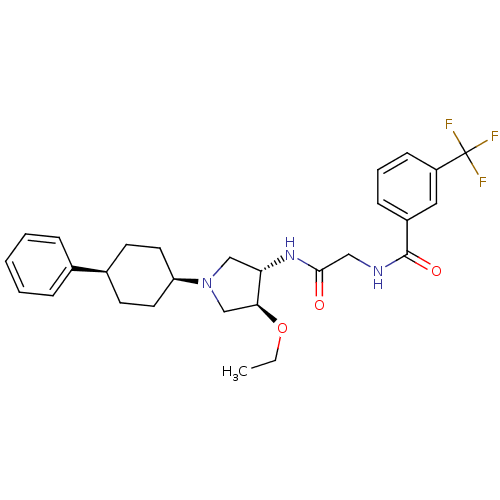
| ||