| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50338522 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_728501 (CHEMBL1685378) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Atkinson, KA; Beretta, EE; Brown, JA; Castrodad, M; Chen, Y; Cosgrove, JM; Du, P; Litchfield, J; Makowski, M; Martin, K; McLellan, TJ; Neagu, C; Perry, DA; Piotrowski, DW; Steppan, CM; Trilles, R N-benzylimidazole carboxamides as potent, orally active stearoylCoA desaturase-1 inhibitors. Bioorg Med Chem Lett21:1621-5 (2011) [PubMed] Article Atkinson, KA; Beretta, EE; Brown, JA; Castrodad, M; Chen, Y; Cosgrove, JM; Du, P; Litchfield, J; Makowski, M; Martin, K; McLellan, TJ; Neagu, C; Perry, DA; Piotrowski, DW; Steppan, CM; Trilles, R N-benzylimidazole carboxamides as potent, orally active stearoylCoA desaturase-1 inhibitors. Bioorg Med Chem Lett21:1621-5 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50338522 |
|---|
| n/a |
|---|
| Name | BDBM50338522 |
|---|
| Synonyms: | 5-methyl-N-(1-methyl-1H-pyrazol-3-yl)-1-(3-(trifluoromethyl)benzyl)-1H-imidazole-4-carboxamide | CHEMBL1683577 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H16F3N5O |
|---|
| Mol. Mass. | 363.337 |
|---|
| SMILES | Cc1c(ncn1Cc1cccc(c1)C(F)(F)F)C(=O)Nc1ccn(C)n1 |
|---|
| Structure | 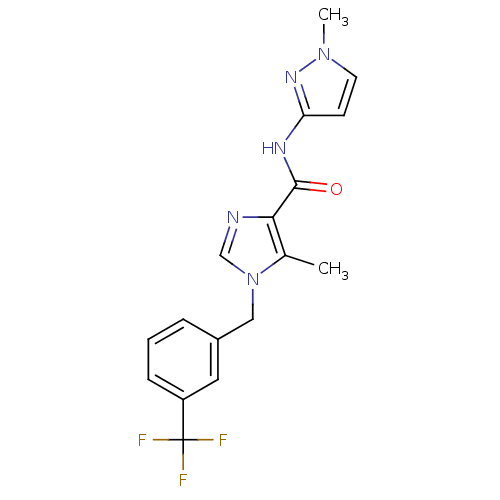
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Atkinson, KA; Beretta, EE; Brown, JA; Castrodad, M; Chen, Y; Cosgrove, JM; Du, P; Litchfield, J; Makowski, M; Martin, K; McLellan, TJ; Neagu, C; Perry, DA; Piotrowski, DW; Steppan, CM; Trilles, R N-benzylimidazole carboxamides as potent, orally active stearoylCoA desaturase-1 inhibitors. Bioorg Med Chem Lett21:1621-5 (2011) [PubMed] Article
Atkinson, KA; Beretta, EE; Brown, JA; Castrodad, M; Chen, Y; Cosgrove, JM; Du, P; Litchfield, J; Makowski, M; Martin, K; McLellan, TJ; Neagu, C; Perry, DA; Piotrowski, DW; Steppan, CM; Trilles, R N-benzylimidazole carboxamides as potent, orally active stearoylCoA desaturase-1 inhibitors. Bioorg Med Chem Lett21:1621-5 (2011) [PubMed] Article