| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine kinase 1 |
|---|
| Ligand | BDBM50312869 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_747639 (CHEMBL1777257) |
|---|
| Ki | 12000±n/a nM |
|---|
| Citation |  Kennedy, AJ; Mathews, TP; Kharel, Y; Field, SD; Moyer, ML; East, JE; Houck, JD; Lynch, KR; Macdonald, TL Development of amidine-based sphingosine kinase 1 nanomolar inhibitors and reduction of sphingosine 1-phosphate in human leukemia cells. J Med Chem54:3524-48 (2011) [PubMed] Article Kennedy, AJ; Mathews, TP; Kharel, Y; Field, SD; Moyer, ML; East, JE; Houck, JD; Lynch, KR; Macdonald, TL Development of amidine-based sphingosine kinase 1 nanomolar inhibitors and reduction of sphingosine 1-phosphate in human leukemia cells. J Med Chem54:3524-48 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine kinase 1 |
|---|
| Name: | Sphingosine kinase 1 |
|---|
| Synonyms: | SK1 | SPHK | SPHK1 | SPHK1_HUMAN | SPK | Sphingosine kinase 1 | Sphingosine kinase 1 (SPHK1) | Sphingosine kinase types 1 (SphK1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 42521.16 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9NYA1 |
|---|
| Residue: | 384 |
|---|
| Sequence: | MDPAGGPRGVLPRPCRVLVLLNPRGGKGKALQLFRSHVQPLLAEAEISFTLMLTERRNHA
RELVRSEELGRWDALVVMSGDGLMHEVVNGLMERPDWETAIQKPLCSLPAGSGNALAASL
NHYAGYEQVTNEDLLTNCTLLLCRRLLSPMNLLSLHTASGLRLFSVLSLAWGFIADVDLE
SEKYRRLGEMRFTLGTFLRLAALRTYRGRLAYLPVGRVGSKTPASPVVVQQGPVDAHLVP
LEEPVPSHWTVVPDEDFVLVLALLHSHLGSEMFAAPMGRCAAGVMHLFYVRAGVSRAMLL
RLFLAMEKGRHMEYECPYLVYVPVVAFRLEPKDGKGVFAVDGELMVSEAVQGQVHPNYFW
MVSGCVEPPPSWKPQQMPPPEEPL
|
|
|
|---|
| BDBM50312869 |
|---|
| n/a |
|---|
| Name | BDBM50312869 |
|---|
| Synonyms: | 4-(4-(4-chlorophenyl)thiazol-2-ylamino)phenol | CHEMBL1076555 | N-aryl-4-aryl-1,3-thiazole-2-amine, 5 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H11ClN2OS |
|---|
| Mol. Mass. | 302.779 |
|---|
| SMILES | Oc1ccc(Nc2nc(cs2)-c2ccc(Cl)cc2)cc1 |
|---|
| Structure | 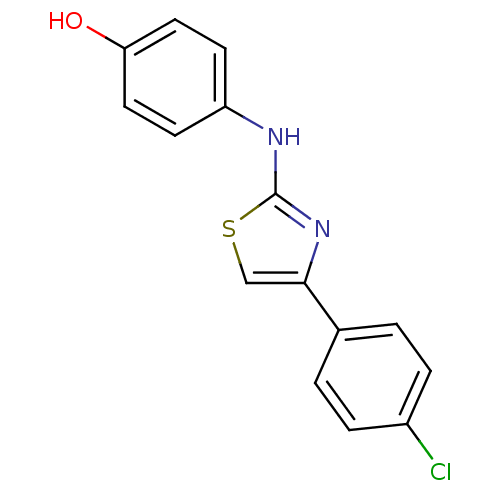
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kennedy, AJ; Mathews, TP; Kharel, Y; Field, SD; Moyer, ML; East, JE; Houck, JD; Lynch, KR; Macdonald, TL Development of amidine-based sphingosine kinase 1 nanomolar inhibitors and reduction of sphingosine 1-phosphate in human leukemia cells. J Med Chem54:3524-48 (2011) [PubMed] Article
Kennedy, AJ; Mathews, TP; Kharel, Y; Field, SD; Moyer, ML; East, JE; Houck, JD; Lynch, KR; Macdonald, TL Development of amidine-based sphingosine kinase 1 nanomolar inhibitors and reduction of sphingosine 1-phosphate in human leukemia cells. J Med Chem54:3524-48 (2011) [PubMed] Article