| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D(2) dopamine receptor |
|---|
| Ligand | BDBM48320 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_61604 |
|---|
| IC50 | 630±n/a nM |
|---|
| Citation |  Kato, S; Morie, T; Kon, T; Yoshida, N; Karasawa, T; Matsumoto, J Novel benzamides as selective and potent gastrokinetic agents. 2. Synthesis and structure-activity relationships of 4-amino-5-chloro-2-ethoxy-N-[[4-(4-fluorobenzyl)-2- morpholinyl]methyl] benzamide citrate (AS-4370) and related compounds. J Med Chem34:616-24 (1991) [PubMed] Kato, S; Morie, T; Kon, T; Yoshida, N; Karasawa, T; Matsumoto, J Novel benzamides as selective and potent gastrokinetic agents. 2. Synthesis and structure-activity relationships of 4-amino-5-chloro-2-ethoxy-N-[[4-(4-fluorobenzyl)-2- morpholinyl]methyl] benzamide citrate (AS-4370) and related compounds. J Med Chem34:616-24 (1991) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D(2) dopamine receptor |
|---|
| Name: | D(2) dopamine receptor |
|---|
| Synonyms: | DOPAMINE D2 | DOPAMINE D2 Long | DOPAMINE D2 Short | DRD2_RAT | Dopamine D2 receptor | Dopamine2-like | Drd2 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 50931.60 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P61169 |
|---|
| Residue: | 444 |
|---|
| Sequence: | MDPLNLSWYDDDLERQNWSRPFNGSEGKADRPHYNYYAMLLTLLIFIIVFGNVLVCMAVS
REKALQTTTNYLIVSLAVADLLVATLVMPWVVYLEVVGEWKFSRIHCDIFVTLDVMMCTA
SILNLCAISIDRYTAVAMPMLYNTRYSSKRRVTVMIAIVWVLSFTISCPLLFGLNNTDQN
ECIIANPAFVVYSSIVSFYVPFIVTLLVYIKIYIVLRKRRKRVNTKRSSRAFRANLKTPL
KGNCTHPEDMKLCTVIMKSNGSFPVNRRRMDAARRAQELEMEMLSSTSPPERTRYSPIPP
SHHQLTLPDPSHHGLHSNPDSPAKPEKNGHAKIVNPRIAKFFEIQTMPNGKTRTSLKTMS
RRKLSQQKEKKATQMLAIVLGVFIICWLPFFITHILNIHCDCNIPPVLYSAFTWLGYVNS
AVNPIIYTTFNIEFRKAFMKILHC
|
|
|
|---|
| BDBM48320 |
|---|
| n/a |
|---|
| Name | BDBM48320 |
|---|
| Synonyms: | 4-amino-5-chloro-N-[2-(diethylamino)ethyl]-2-methoxy-benzamide;hydrochloride | 4-amino-5-chloro-N-[2-(diethylamino)ethyl]-2-methoxybenzamide;hydrochloride | 4-azanyl-5-chloranyl-N-[2-(diethylamino)ethyl]-2-methoxy-benzamide;hydrochloride | METOCLOPRAMIDE | METOCLOPRAMIDE HYDROCHLORIDE | MLS000069667 | Metochloropramide | SMR000058471 | US11147820, Compound Metoclopramide | US11717500, Compound Metoclopramide | US9132134, Metoclopramide | cid_23659 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H22ClN3O2 |
|---|
| Mol. Mass. | 299.796 |
|---|
| SMILES | CCN(CC)CCNC(=O)c1cc(Cl)c(N)cc1OC |
|---|
| Structure | 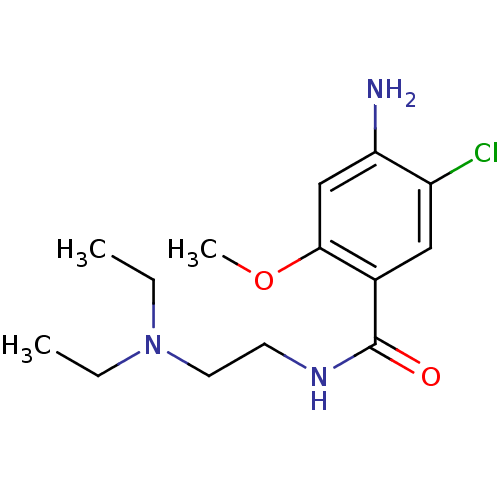
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kato, S; Morie, T; Kon, T; Yoshida, N; Karasawa, T; Matsumoto, J Novel benzamides as selective and potent gastrokinetic agents. 2. Synthesis and structure-activity relationships of 4-amino-5-chloro-2-ethoxy-N-[[4-(4-fluorobenzyl)-2- morpholinyl]methyl] benzamide citrate (AS-4370) and related compounds. J Med Chem34:616-24 (1991) [PubMed]
Kato, S; Morie, T; Kon, T; Yoshida, N; Karasawa, T; Matsumoto, J Novel benzamides as selective and potent gastrokinetic agents. 2. Synthesis and structure-activity relationships of 4-amino-5-chloro-2-ethoxy-N-[[4-(4-fluorobenzyl)-2- morpholinyl]methyl] benzamide citrate (AS-4370) and related compounds. J Med Chem34:616-24 (1991) [PubMed]