| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1D |
|---|
| Ligand | BDBM50033434 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2035 (CHEMBL616869) |
|---|
| Ki | 0.39±n/a nM |
|---|
| Citation |  Perez, M; Fourrier, C; Sigogneau, I; Pauwels, PJ; Palmier, C; John, GW; Valentin, JP; Halazy, S Synthesis and serotonergic activity of arylpiperazide derivatives of serotonin: potent agonists for 5-HT1D receptors. J Med Chem38:3602-7 (1995) [PubMed] Perez, M; Fourrier, C; Sigogneau, I; Pauwels, PJ; Palmier, C; John, GW; Valentin, JP; Halazy, S Synthesis and serotonergic activity of arylpiperazide derivatives of serotonin: potent agonists for 5-HT1D receptors. J Med Chem38:3602-7 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1D |
|---|
| Name: | 5-hydroxytryptamine receptor 1D |
|---|
| Synonyms: | 5-HT-1D | 5-HT-1D-alpha | 5-HT1D | 5-hydroxytryptamine receptor 1D (5-HT1D) | 5HT1D_HUMAN | HTR1D | HTR1DA | HTRL | Serotonin (5-HT) receptor | Serotonin Receptor 1D |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 41920.63 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells. |
|---|
| Residue: | 377 |
|---|
| Sequence: | MSPLNQSAEGLPQEASNRSLNATETSEAWDPRTLQALKISLAVVLSVITLATVLSNAFVL
TTILLTRKLHTPANYLIGSLATTDLLVSILVMPISIAYTITHTWNFGQILCDIWLSSDIT
CCTASILHLCVIALDRYWAITDALEYSKRRTAGHAATMIAIVWAISICISIPPLFWRQAK
AQEEMSDCLVNTSQISYTIYSTCGAFYIPSVLLIILYGRIYRAARNRILNPPSLYGKRFT
TAHLITGSAGSSLCSLNSSLHEGHSHSAGSPLFFNHVKIKLADSALERKRISAARERKAT
KILGIILGAFIICWLPFFVVSLVLPICRDSCWIHPALFDFFTWLGYLNSLINPIIYTVFN
EEFRQAFQKIVPFRKAS
|
|
|
|---|
| BDBM50033434 |
|---|
| n/a |
|---|
| Name | BDBM50033434 |
|---|
| Synonyms: | 2-(4-{2-[3-(2-Amino-ethyl)-1H-indol-5-yloxy]-acetyl}-piperazin-1-yl)-benzonitrile | CHEMBL119927 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25N5O2 |
|---|
| Mol. Mass. | 403.4769 |
|---|
| SMILES | NCCc1c[nH]c2ccc(OCC(=O)N3CCN(CC3)c3ccccc3C#N)cc12 |
|---|
| Structure | 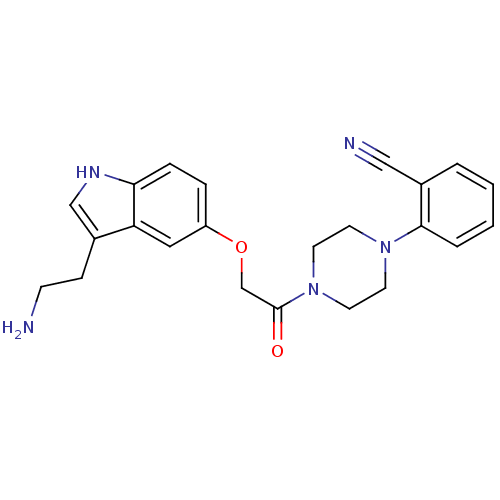
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Perez, M; Fourrier, C; Sigogneau, I; Pauwels, PJ; Palmier, C; John, GW; Valentin, JP; Halazy, S Synthesis and serotonergic activity of arylpiperazide derivatives of serotonin: potent agonists for 5-HT1D receptors. J Med Chem38:3602-7 (1995) [PubMed]
Perez, M; Fourrier, C; Sigogneau, I; Pauwels, PJ; Palmier, C; John, GW; Valentin, JP; Halazy, S Synthesis and serotonergic activity of arylpiperazide derivatives of serotonin: potent agonists for 5-HT1D receptors. J Med Chem38:3602-7 (1995) [PubMed]