| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sodium-dependent noradrenaline transporter |
|---|
| Ligand | BDBM50084338 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_140881 (CHEMBL752512) |
|---|
| Ki | 150±n/a nM |
|---|
| Citation |  Choi, SW; Elmaleh, DR; Hanson, RN; Fischman, AJ Novel 3-aminomethyl- and 4-aminopiperidine analogues of 1-[2-(diphenylmethoxy)ethyl]-4-(3-phenylpropyl)piperazines: synthesis and evaluation as dopamine transporter ligands. J Med Chem43:205-13 (2000) [PubMed] Choi, SW; Elmaleh, DR; Hanson, RN; Fischman, AJ Novel 3-aminomethyl- and 4-aminopiperidine analogues of 1-[2-(diphenylmethoxy)ethyl]-4-(3-phenylpropyl)piperazines: synthesis and evaluation as dopamine transporter ligands. J Med Chem43:205-13 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sodium-dependent noradrenaline transporter |
|---|
| Name: | Sodium-dependent noradrenaline transporter |
|---|
| Synonyms: | Monoamine transporter | NAT1 | NET | NET1 | Noradrenaline Transporter (NET) | Norepinephrine Transporter (NET) | Norepinephrine transporter protein (NET) | SC6A2_HUMAN | SLC6A2 | SLC6A5 | Sodium-dependent noradrenaline transporter (NET) | Solute carrier family 6 member 2 |
|---|
| Type: | Multi-pass membrane protein |
|---|
| Mol. Mass.: | 69337.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P23975 |
|---|
| Residue: | 617 |
|---|
| Sequence: | MLLARMNPQVQPENNGADTGPEQPLRARKTAELLVVKERNGVQCLLAPRDGDAQPRETWG
KKIDFLLSVVGFAVDLANVWRFPYLCYKNGGGAFLIPYTLFLIIAGMPLFYMELALGQYN
REGAATVWKICPFFKGVGYAVILIALYVGFYYNVIIAWSLYYLFSSFTLNLPWTDCGHTW
NSPNCTDPKLLNGSVLGNHTKYSKYKFTPAAEFYERGVLHLHESSGIHDIGLPQWQLLLC
LMVVVIVLYFSLWKGVKTSGKVVWITATLPYFVLFVLLVHGVTLPGASNGINAYLHIDFY
RLKEATVWIDAATQIFFSLGAGFGVLIAFASYNKFDNNCYRDALLTSSINCITSFVSGFA
IFSILGYMAHEHKVNIEDVATEGAGLVFILYPEAISTLSGSTFWAVVFFVMLLALGLDSS
MGGMEAVITGLADDFQVLKRHRKLFTFGVTFSTFLLALFCITKGGIYVLTLLDTFAAGTS
ILFAVLMEAIGVSWFYGVDRFSNDIQQMMGFRPGLYWRLCWKFVSPAFLLFVVVVSIINF
KPLTYDDYIFPPWANWVGWGIALSSMVLVPIYVIYKFLSTQGSLWERLAYGITPENEHHL
VAQRDIRQFQLQHWLAI
|
|
|
|---|
| BDBM50084338 |
|---|
| n/a |
|---|
| Name | BDBM50084338 |
|---|
| Synonyms: | (2-Benzhydryloxy-ethyl)-{1-[2-(4-fluoro-phenyl)-ethyl]-piperidin-3-ylmethyl}-amine | CHEMBL66112 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H35FN2O |
|---|
| Mol. Mass. | 446.5994 |
|---|
| SMILES | Fc1ccc(CCN2CCCC(CNCCOC(c3ccccc3)c3ccccc3)C2)cc1 |
|---|
| Structure | 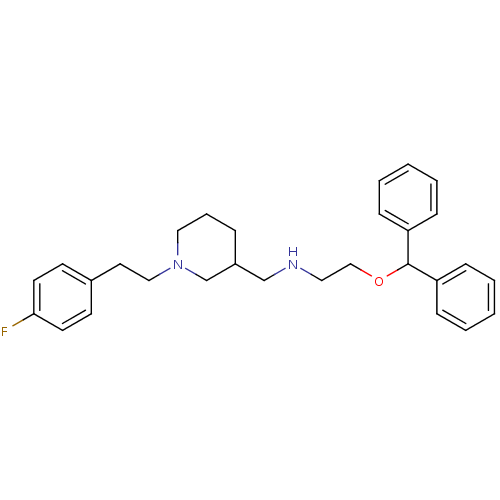
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Choi, SW; Elmaleh, DR; Hanson, RN; Fischman, AJ Novel 3-aminomethyl- and 4-aminopiperidine analogues of 1-[2-(diphenylmethoxy)ethyl]-4-(3-phenylpropyl)piperazines: synthesis and evaluation as dopamine transporter ligands. J Med Chem43:205-13 (2000) [PubMed]
Choi, SW; Elmaleh, DR; Hanson, RN; Fischman, AJ Novel 3-aminomethyl- and 4-aminopiperidine analogues of 1-[2-(diphenylmethoxy)ethyl]-4-(3-phenylpropyl)piperazines: synthesis and evaluation as dopamine transporter ligands. J Med Chem43:205-13 (2000) [PubMed]