| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 5A |
|---|
| Ligand | BDBM50005835 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1079 (CHEMBL616402) |
|---|
| IC50 | 501±n/a nM |
|---|
| Citation |  Castro, JL; Baker, R; Guiblin, AR; Hobbs, SC; Jenkins, MR; Russell, MG; Beer, MS; Stanton, JA; Scholey, K; Hargreaves, RJ Synthesis and biological activity of 3-[2-(dimethylamino)ethyl]-5-[(1,1-dioxo-5-methyl-1,2,5-thiadiazolidin- 2-yl)-methyl]-1H-indole and analogues: agonists for the 5-HT1D receptor. J Med Chem37:3023-32 (1994) [PubMed] Castro, JL; Baker, R; Guiblin, AR; Hobbs, SC; Jenkins, MR; Russell, MG; Beer, MS; Stanton, JA; Scholey, K; Hargreaves, RJ Synthesis and biological activity of 3-[2-(dimethylamino)ethyl]-5-[(1,1-dioxo-5-methyl-1,2,5-thiadiazolidin- 2-yl)-methyl]-1H-indole and analogues: agonists for the 5-HT1D receptor. J Med Chem37:3023-32 (1994) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 5A |
|---|
| Name: | 5-hydroxytryptamine receptor 5A |
|---|
| Synonyms: | 5-HT-5 | 5-HT-5A | 5-hydroxytryptamine receptor 5 (5-HT5) | 5-hydroxytryptamine receptor 5A (5-HT5A) | 5HT5A_HUMAN | HTR5A | Serotonin (5-HT) receptor | Serotonin receptor 5A |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40266.25 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P47898 |
|---|
| Residue: | 357 |
|---|
| Sequence: | MDLPVNLTSFSLSTPSPLETNHSLGKDDLRPSSPLLSVFGVLILTLLGFLVAATFAWNLL
VLATILRVRTFHRVPHNLVASMAVSDVLVAALVMPLSLVHELSGRRWQLGRRLCQLWIAC
DVLCCTASIWNVTAIALDRYWSITRHMEYTLRTRKCVSNVMIALTWALSAVISLAPLLFG
WGETYSEGSEECQVSREPSYAVFSTVGAFYLPLCVVLFVYWKIYKAAKFRVGSRKTNSVS
PISEAVEVKDSAKQPQMVFTVRHATVTFQPEGDTWREQKEQRAALMVGILIGVFVLCWIP
FFLTELISPLCSCDIPAIWKSIFLWLGYSNSFFNPLIYTAFNKNYNSAFKNFFSRQH
|
|
|
|---|
| BDBM50005835 |
|---|
| n/a |
|---|
| Name | BDBM50005835 |
|---|
| Synonyms: | (3-[2-(dimethylamino)ethyl]-1H-indol-5-yl)-N-methylmethanesulfonamide | 1-[3-(2-dimethylaminoethyl)-1H-indol-5-yl]-N-methyl-methanesulfonamide | 1-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide | 3-(2-(dimethylamino)ethyl)-N-methyl-1H-indole-5-methanesulfonamide | 3-[2-(dimethylamino)ethyl]-N-methylindole-5-methanesulfonamide | CHEMBL128 | SUMATRIPTAN | Sumatran | Sumax |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H21N3O2S |
|---|
| Mol. Mass. | 295.4 |
|---|
| SMILES | CNS(=O)(=O)Cc1ccc2[nH]cc(CCN(C)C)c2c1 |
|---|
| Structure | 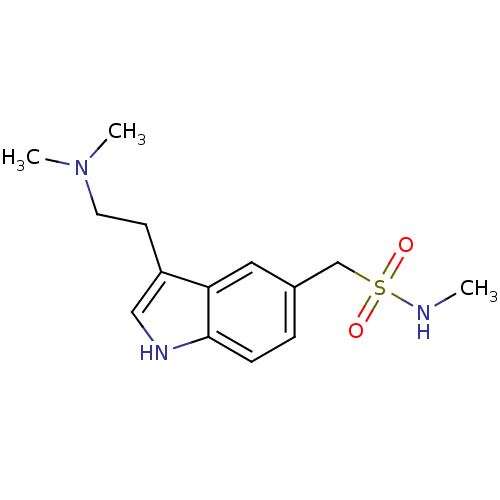
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Castro, JL; Baker, R; Guiblin, AR; Hobbs, SC; Jenkins, MR; Russell, MG; Beer, MS; Stanton, JA; Scholey, K; Hargreaves, RJ Synthesis and biological activity of 3-[2-(dimethylamino)ethyl]-5-[(1,1-dioxo-5-methyl-1,2,5-thiadiazolidin- 2-yl)-methyl]-1H-indole and analogues: agonists for the 5-HT1D receptor. J Med Chem37:3023-32 (1994) [PubMed]
Castro, JL; Baker, R; Guiblin, AR; Hobbs, SC; Jenkins, MR; Russell, MG; Beer, MS; Stanton, JA; Scholey, K; Hargreaves, RJ Synthesis and biological activity of 3-[2-(dimethylamino)ethyl]-5-[(1,1-dioxo-5-methyl-1,2,5-thiadiazolidin- 2-yl)-methyl]-1H-indole and analogues: agonists for the 5-HT1D receptor. J Med Chem37:3023-32 (1994) [PubMed]