| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50431828 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_952207 (CHEMBL2351003) |
|---|
| IC50 | 13600±n/a nM |
|---|
| Citation |  Jeyachandran, V; Kumar, RR; Ali, MA; Choon, TS A one-pot domino synthesis and discovery of highly functionalized dihydrobenzo[b]thiophenes as AChE inhibitors. Bioorg Med Chem Lett23:2101-5 (2013) [PubMed] Article Jeyachandran, V; Kumar, RR; Ali, MA; Choon, TS A one-pot domino synthesis and discovery of highly functionalized dihydrobenzo[b]thiophenes as AChE inhibitors. Bioorg Med Chem Lett23:2101-5 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_ELEEL | Acetylcholinesterase (AChE) | Acetylcholinesterase (EeAChE) | ache |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 71812.79 |
|---|
| Organism: | Electrophorus electricus (Electric eel) |
|---|
| Description: | n/a |
|---|
| Residue: | 633 |
|---|
| Sequence: | MKILDALLFPVIFIMFFIHLSIAQTDPELTIMTRLGQVQGTRLPVPDRSHVIAFLGIPFA
EPPLGKMRFKPPEPKKPWNDVFDARDYPSACYQYVDTSYPGFSGTEMWNPNRMMSEDCLY
LNVWVPATPRPHNLTVMVWIYGGGFYSGSSSLDVYDGRYLAHSEKVVVVSMNYRVSAFGF
LALNGSAEAPGNVGLLDQRLALQWVQDNIHFFGGNPKQVTIFGESAGAASVGMHLLSPDS
RPKFTRAILQSGVPNGPWRTVSFDEARRRAIKLGRLVGCPDGNDTDLIDCLRSKQPQDLI
DQEWLVLPFSGLFRFSFVPVIDGVVFPDTPEAMLNSGNFKDTQILLGVNQNEGSYFLIYG
APGFSKDNESLITREDFLQGVKMSVPHANEIGLEAVILQYTDWMDEDNPIKNREAMDDIV
GDHNVVCPLQHFAKMYAQYSILQGQTGTASQGNLGWGNSGSASNSGNSQVSVYLYMFDHR
ASNLVWPEWMGVIHGYEIEFVFGLPLEKRLNYTLEEEKLSRRMMKYWANFARTGNPNINV
DGSIDSRRRWPVFTSTEQKHVGLNTDSLKVHKGLKSQFCALWNRFLPRLLNVTENIDDAE
RQWKAEFHRWSSYMMHWKNQFDHYSKQERCTNL
|
|
|
|---|
| BDBM50431828 |
|---|
| n/a |
|---|
| Name | BDBM50431828 |
|---|
| Synonyms: | CHEMBL2347094 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H16ClN3S |
|---|
| Mol. Mass. | 401.911 |
|---|
| SMILES | Cc1ccc(cc1)C1Cc2c(S1)c(-c1ccc(Cl)cc1)c(C#N)c(N)c2C#N |
|---|
| Structure | 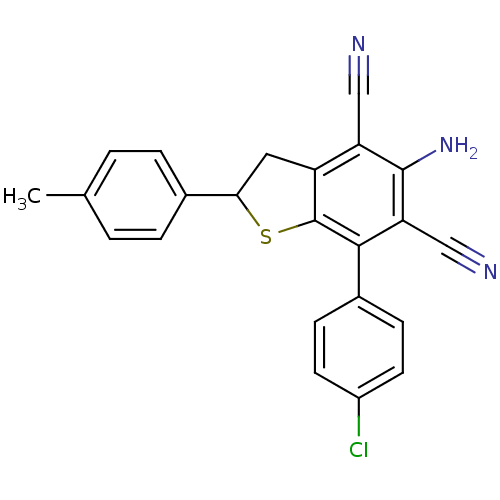
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jeyachandran, V; Kumar, RR; Ali, MA; Choon, TS A one-pot domino synthesis and discovery of highly functionalized dihydrobenzo[b]thiophenes as AChE inhibitors. Bioorg Med Chem Lett23:2101-5 (2013) [PubMed] Article
Jeyachandran, V; Kumar, RR; Ali, MA; Choon, TS A one-pot domino synthesis and discovery of highly functionalized dihydrobenzo[b]thiophenes as AChE inhibitors. Bioorg Med Chem Lett23:2101-5 (2013) [PubMed] Article