| Reaction Details |
|---|
 | Report a problem with these data |
| Target | G-protein coupled receptor 35 |
|---|
| Ligand | BDBM50185 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_983065 (CHEMBL2427425) |
|---|
| IC50 | 27±n/a nM |
|---|
| Citation |  Thimm, D; Funke, M; Meyer, A; Müller, CE 6-Bromo-8-(4-[(3)H]methoxybenzamido)-4-oxo-4H-chromene-2-carboxylic Acid: a powerful tool for studying orphan G protein-coupled receptor GPR35. J Med Chem56:7084-99 (2013) [PubMed] Article Thimm, D; Funke, M; Meyer, A; Müller, CE 6-Bromo-8-(4-[(3)H]methoxybenzamido)-4-oxo-4H-chromene-2-carboxylic Acid: a powerful tool for studying orphan G protein-coupled receptor GPR35. J Med Chem56:7084-99 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| G-protein coupled receptor 35 |
|---|
| Name: | G-protein coupled receptor 35 |
|---|
| Synonyms: | G protein-coupled receptor 35 | GPR35 | GPR35_HUMAN |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 34085.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_33695097 |
|---|
| Residue: | 309 |
|---|
| Sequence: | MNGTYNTCGSSDLTWPPAIKLGFYAYLGVLLVLGLLLNSLALWVFCCRMQQWTETRIYMT
NLAVADLCLLCTLPFVLHSLRDTSDTPLCQLSQGIYLTNRYMSISLVTAIAVDRYVAVRH
PLRARGLRSPRQAAAVCAVLWVLVIGSLVARWLLGIQEGGFCFRSTRHNFNSMAFPLLGF
YLPLAVVVFCSLKVVTALAQRPPTDVGQAEATRKAARMVWANLLVFVVCFLPLHVGLTVR
LAVGWNACALLETIRRALYITSKLSDANCCLDAICYYYMAKEFQEASALAVAPSAKAHKS
QDSLCVTLA
|
|
|
|---|
| BDBM50185 |
|---|
| n/a |
|---|
| Name | BDBM50185 |
|---|
| Synonyms: | 2-hydroxy-4-({4-[5-(2-methyl-3-phenyl-2-propen-1-ylidene)-4-oxo-2-thioxo-1,3-thiazolidin-3-yl]butanoyl}amino)benzoic acid | 2-hydroxy-4-[4-[(5Z)-4-keto-5-[(E)-2-methyl-3-phenyl-prop-2-enylidene]-2-thioxo-thiazolidin-3-yl]butanoylamino]benzoic acid | 2-hydroxy-4-[4-[(5Z)-5-[(E)-2-methyl-3-phenylprop-2-enylidene]-4-oxo-2-sulfanylidene-1,3-thiazolidin-3-yl]butanoylamino]benzoic acid | 2-hydroxy-4-[[4-[(5Z)-5-[(E)-2-methyl-3-phenylprop-2-enylidene]-4-oxo-2-sulfanylidene-3-thiazolidinyl]-1-oxobutyl]amino]benzoic acid | 4-[4-[(5Z)-5-[(E)-2-methyl-3-phenyl-prop-2-enylidene]-4-oxidanylidene-2-sulfanylidene-1,3-thiazolidin-3-yl]butanoylamino]-2-oxidanyl-benzoic acid | CHEMBL1384502 | MLS000575039 | SMR000208964 | cid_2286812 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H22N2O5S2 |
|---|
| Mol. Mass. | 482.572 |
|---|
| SMILES | C\C(\C=C1/SC(=S)N(CCCC(=O)Nc2ccc(C(O)=O)c(O)c2)C1=O)=C/c1ccccc1 |
|---|
| Structure | 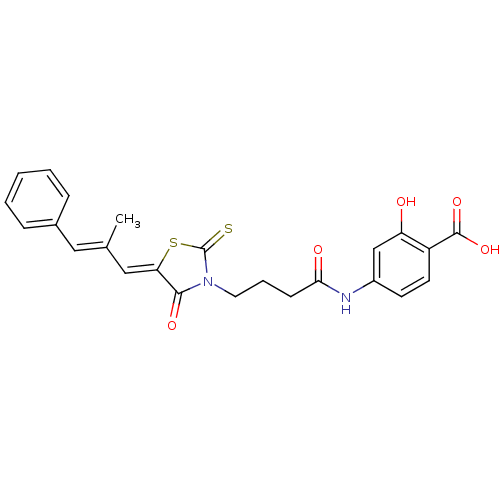
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Thimm, D; Funke, M; Meyer, A; Müller, CE 6-Bromo-8-(4-[(3)H]methoxybenzamido)-4-oxo-4H-chromene-2-carboxylic Acid: a powerful tool for studying orphan G protein-coupled receptor GPR35. J Med Chem56:7084-99 (2013) [PubMed] Article
Thimm, D; Funke, M; Meyer, A; Müller, CE 6-Bromo-8-(4-[(3)H]methoxybenzamido)-4-oxo-4H-chromene-2-carboxylic Acid: a powerful tool for studying orphan G protein-coupled receptor GPR35. J Med Chem56:7084-99 (2013) [PubMed] Article