| Reaction Details |
|---|
 | Report a problem with these data |
| Target | DNA topoisomerase 1 |
|---|
| Ligand | BDBM50332532 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1520827 (CHEMBL3626095) |
|---|
| EC50 | >100000±n/a nM |
|---|
| Citation |  Majumdar, P; Bathula, C; Basu, SM; Das, SK; Agarwal, R; Hati, S; Singh, A; Sen, S; Das, BB Design, synthesis and evaluation of thiohydantoin derivatives as potent topoisomerase I (Top1) inhibitors with anticancer activity. Eur J Med Chem102:540-51 (2015) [PubMed] Article Majumdar, P; Bathula, C; Basu, SM; Das, SK; Agarwal, R; Hati, S; Singh, A; Sen, S; Das, BB Design, synthesis and evaluation of thiohydantoin derivatives as potent topoisomerase I (Top1) inhibitors with anticancer activity. Eur J Med Chem102:540-51 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| DNA topoisomerase 1 |
|---|
| Name: | DNA topoisomerase 1 |
|---|
| Synonyms: | DNA topoisomerase I | DNA topoisomerase I (Topo I) | TOP1 | TOP1_HUMAN |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 90771.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11387 |
|---|
| Residue: | 765 |
|---|
| Sequence: | MSGDHLHNDSQIEADFRLNDSHKHKDKHKDREHRHKEHKKEKDREKSKHSNSEHKDSEKK
HKEKEKTKHKDGSSEKHKDKHKDRDKEKRKEEKVRASGDAKIKKEKENGFSSPPQIKDEP
EDDGYFVPPKEDIKPLKRPRDEDDADYKPKKIKTEDTKKEKKRKLEEEEDGKLKKPKNKD
KDKKVPEPDNKKKKPKKEEEQKWKWWEEERYPEGIKWKFLEHKGPVFAPPYEPLPENVKF
YYDGKVMKLSPKAEEVATFFAKMLDHEYTTKEIFRKNFFKDWRKEMTNEEKNIITNLSKC
DFTQMSQYFKAQTEARKQMSKEEKLKIKEENEKLLKEYGFCIMDNHKERIANFKIEPPGL
FRGRGNHPKMGMLKRRIMPEDIIINCSKDAKVPSPPPGHKWKEVRHDNKVTWLVSWTENI
QGSIKYIMLNPSSRIKGEKDWQKYETARRLKKCVDKIRNQYREDWKSKEMKVRQRAVALY
FIDKLALRAGNEKEEGETADTVGCCSLRVEHINLHPELDGQEYVVEFDFLGKDSIRYYNK
VPVEKRVFKNLQLFMENKQPEDDLFDRLNTGILNKHLQDLMEGLTAKVFRTYNASITLQQ
QLKELTAPDENIPAKILSYNRANRAVAILCNHQRAPPKTFEKSMMNLQTKIDAKKEQLAD
ARRDLKSAKADAKVMKDAKTKKVVESKKKAVQRLEEQLMKLEVQATDREENKQIALGTSK
LNYLDPRITVAWCKKWGVPIEKIYNKTQREKFAWAIDMADEDYEF
|
|
|
|---|
| BDBM50332532 |
|---|
| n/a |
|---|
| Name | BDBM50332532 |
|---|
| Synonyms: | (E)-5-(3,4,5-trimethoxybenzylidene)imidazolidine-2,4-dione | CHEMBL1630442 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H14N2O5 |
|---|
| Mol. Mass. | 278.2607 |
|---|
| SMILES | COc1cc(\C=C2\NC(=O)NC2=O)cc(OC)c1OC |
|---|
| Structure | 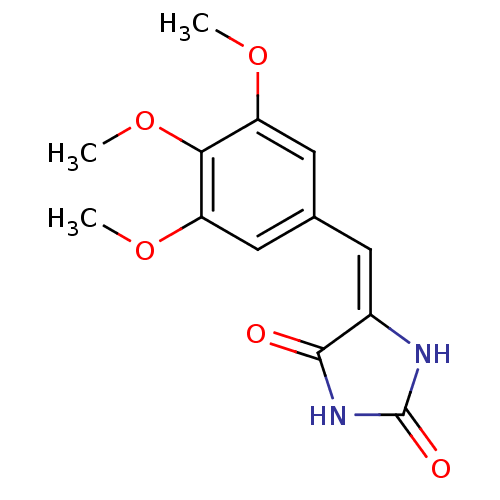
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Majumdar, P; Bathula, C; Basu, SM; Das, SK; Agarwal, R; Hati, S; Singh, A; Sen, S; Das, BB Design, synthesis and evaluation of thiohydantoin derivatives as potent topoisomerase I (Top1) inhibitors with anticancer activity. Eur J Med Chem102:540-51 (2015) [PubMed] Article
Majumdar, P; Bathula, C; Basu, SM; Das, SK; Agarwal, R; Hati, S; Singh, A; Sen, S; Das, BB Design, synthesis and evaluation of thiohydantoin derivatives as potent topoisomerase I (Top1) inhibitors with anticancer activity. Eur J Med Chem102:540-51 (2015) [PubMed] Article