| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Oxysterols receptor LXR-beta |
|---|
| Ligand | BDBM50172720 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1583942 (CHEMBL3815374) |
|---|
| EC50 | 800±n/a nM |
|---|
| Citation |  Koura, M; Yamaguchi, Y; Kurobuchi, S; Sumida, H; Watanabe, Y; Enomoto, T; Matsuda, T; Okuda, A; Koshizawa, T; Matsumoto, Y; Shibuya, K Discovery of a 2-hydroxyacetophenone derivative as an outstanding linker to enhance potency and�-selectivity of liver X receptor agonist. Bioorg Med Chem24:3436-46 (2016) [PubMed] Article Koura, M; Yamaguchi, Y; Kurobuchi, S; Sumida, H; Watanabe, Y; Enomoto, T; Matsuda, T; Okuda, A; Koshizawa, T; Matsumoto, Y; Shibuya, K Discovery of a 2-hydroxyacetophenone derivative as an outstanding linker to enhance potency and�-selectivity of liver X receptor agonist. Bioorg Med Chem24:3436-46 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Oxysterols receptor LXR-beta |
|---|
| Name: | Oxysterols receptor LXR-beta |
|---|
| Synonyms: | LXRB | Liver X receptor beta (NR1H2) | Liver X, LXR beta | NER | NR1H2 | NR1H2_HUMAN | Nuclear receptor NER | UNR | Ubiquitously-expressed nuclear receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 50978.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P55055 |
|---|
| Residue: | 460 |
|---|
| Sequence: | MSSPTTSSLDTPLPGNGPPQPGAPSSSPTVKEEGPEPWPGGPDPDVPGTDEASSACSTDW
VIPDPEEEPERKRKKGPAPKMLGHELCRVCGDKASGFHYNVLSCEGCKGFFRRSVVRGGA
RRYACRGGGTCQMDAFMRRKCQQCRLRKCKEAGMREQCVLSEEQIRKKKIRKQQQESQSQ
SQSPVGPQGSSSSASGPGASPGGSEAGSQGSGEGEGVQLTAAQELMIQQLVAAQLQCNKR
SFSDQPKVTPWPLGADPQSRDARQQRFAHFTELAIISVQEIVDFAKQVPGFLQLGREDQI
ALLKASTIEIMLLETARRYNHETECITFLKDFTYSKDDFHRAGLQVEFINPIFEFSRAMR
RLGLDDAEYALLIAINIFSADRPNVQEPGRVEALQQPYVEALLSYTRIKRPQDQLRFPRM
LMKLVSLRTLSSVHSEQVFALRLQDKKLPPLLSEIWDVHE
|
|
|
|---|
| BDBM50172720 |
|---|
| n/a |
|---|
| Name | BDBM50172720 |
|---|
| Synonyms: | CHEMBL3814017 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H38F6N2O5 |
|---|
| Mol. Mass. | 644.6449 |
|---|
| SMILES | CCCc1cc(cc(CCC)c1OC\C=C\CN1C(=O)NC(C)(C1=O)c1ccc(OC(C)C)cc1)C(O)(C(F)(F)F)C(F)(F)F |
|---|
| Structure | 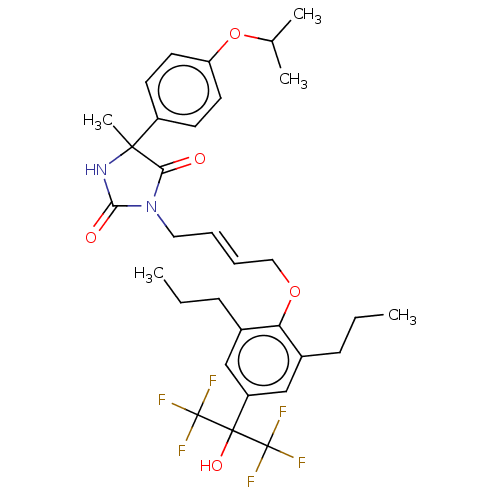
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Koura, M; Yamaguchi, Y; Kurobuchi, S; Sumida, H; Watanabe, Y; Enomoto, T; Matsuda, T; Okuda, A; Koshizawa, T; Matsumoto, Y; Shibuya, K Discovery of a 2-hydroxyacetophenone derivative as an outstanding linker to enhance potency and�-selectivity of liver X receptor agonist. Bioorg Med Chem24:3436-46 (2016) [PubMed] Article
Koura, M; Yamaguchi, Y; Kurobuchi, S; Sumida, H; Watanabe, Y; Enomoto, T; Matsuda, T; Okuda, A; Koshizawa, T; Matsumoto, Y; Shibuya, K Discovery of a 2-hydroxyacetophenone derivative as an outstanding linker to enhance potency and�-selectivity of liver X receptor agonist. Bioorg Med Chem24:3436-46 (2016) [PubMed] Article