| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Gamma-aminobutyric acid receptor subunit alpha-1 |
|---|
| Ligand | BDBM50019213 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 18.6±n/a nM |
|---|
| Comments | PDSP_543 |
|---|
| Citation |  Yamagishi, H; Kawaguchi, M Characterization of central- and peripheral-type benzodiazepine receptors in rat salivary glands. Biochem Pharmacol55:209-14 (1998) [PubMed] Article Yamagishi, H; Kawaguchi, M Characterization of central- and peripheral-type benzodiazepine receptors in rat salivary glands. Biochem Pharmacol55:209-14 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Gamma-aminobutyric acid receptor subunit alpha-1 |
|---|
| Name: | Gamma-aminobutyric acid receptor subunit alpha-1 |
|---|
| Synonyms: | Benzodiazepine central | Benzodiazepine receptors | GABA A Benzodiazepine | GABA A Benzodiazepine Type I | GABA A Benzodiazepine Type II | GABA A Benzodiazepine Type IIL | GABA A Benzodiazepine Type IIM | GABA A Benzodiazepine omega1 | GABA A Benzodiazepine omega2 | GABA A Benzodiazepine omega5 | GABA A alpha1 | GABA A anti-Alpha1 | GABA receptor alpha-1 subunit | GABA, Chloride, TBOB | GABA-PICROTOXIN | GBRA1_RAT | Gabra-1 | Gabra1 | TBPS |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51770.21 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | P62813 |
|---|
| Residue: | 455 |
|---|
| Sequence: | MKKSRGLSDYLWAWTLILSTLSGRSYGQPSQDELKDNTTVFTRILDRLLDGYDNRLRPGL
GERVTEVKTDIFVTSFGPVSDHDMEYTIDVFFRQSWKDERLKFKGPMTVLRLNNLMASKI
WTPDTFFHNGKKSVAHNMTMPNKLLRITEDGTLLYTMRLTVRAECPMHLEDFPMDAHACP
LKFGSYAYTRAEVVYEWTREPARSVVVAEDGSRLNQYDLLGQTVDSGIVQSSTGEYVVMT
THFHLKRKIGYFVIQTYLPCIMTVILSQVSFWLNRESVPARTVFGVTTVLTMTTLSISAR
NSLPKVAYATAMDWFIAVCYAFVFSALIEFATVNYFTKRGYAWDGKSVVPEKPKKVKDPL
IKKNNTYAPTATSYTPNLARGDPGLATIAKSATIEPKEVKPETKPPEPKKTFNSVSKIDR
LSRIAFPLLFGIFNLVYWATYLNREPQLKAPTPHQ
|
|
|
|---|
| BDBM50019213 |
|---|
| n/a |
|---|
| Name | BDBM50019213 |
|---|
| Synonyms: | 1,3-dihydro-7-nitro-5-(2-chlorophenyl)-2H-1,4.benzodiazepin-2-one | 5-(2-chlorophenyl)-7-nitro-1,3-dihydro-2H-1,4-benzodiazepin-2-one | 5-(o-chlorophenyl)-7-nitro-1,3-dihydro-2H-1,4-benzodiazepin-2-one | CHEMBL452 | CLONAZEPAM |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H10ClN3O3 |
|---|
| Mol. Mass. | 315.711 |
|---|
| SMILES | [O-][N+](=O)c1ccc2NC(=O)CN=C(c3ccccc3Cl)c2c1 |t:11| |
|---|
| Structure | 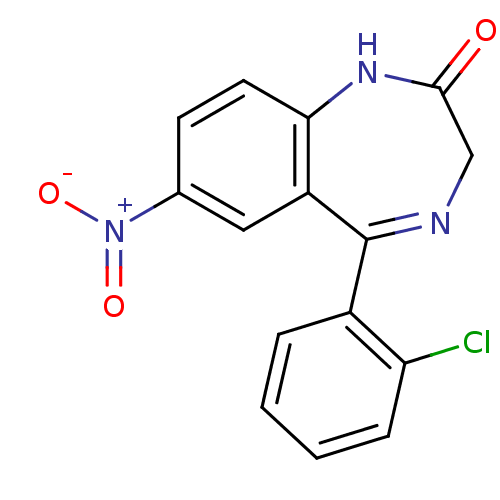
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yamagishi, H; Kawaguchi, M Characterization of central- and peripheral-type benzodiazepine receptors in rat salivary glands. Biochem Pharmacol55:209-14 (1998) [PubMed] Article
Yamagishi, H; Kawaguchi, M Characterization of central- and peripheral-type benzodiazepine receptors in rat salivary glands. Biochem Pharmacol55:209-14 (1998) [PubMed] Article