

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A | ||
| Ligand | BDBM144672 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | IMAP TR-FRET Assay | ||
| Temperature | 298.15±n/a K | ||
| IC50 | 4.11±n/a nM | ||
| Comments | extracted | ||
| Citation |  Allen, JR; Frohn, MJ; Harrington, PE; Hu, E; Pickrell, AJ; Rzasa, RM; Sham, KK Heteroaryloxycarbocyclyl compounds as PDE10 inhibitors US Patent US8952037 Publication Date 2/10/2015 Allen, JR; Frohn, MJ; Harrington, PE; Hu, E; Pickrell, AJ; Rzasa, RM; Sham, KK Heteroaryloxycarbocyclyl compounds as PDE10 inhibitors US Patent US8952037 Publication Date 2/10/2015 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A | |||
| Name: | cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A | ||
| Synonyms: | 3',5'-cyclic phosphodiesterase | 3.1.4.17 | PDE10A | PDE10_HUMAN | Phosphodiesterase 10 (PDE10) | Phosphodiesterase 10A | ||
| Type: | Protein | ||
| Mol. Mass.: | 88412.52 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Q9Y233 | ||
| Residue: | 1055 | ||
| Sequence: |
| ||
| BDBM144672 | |||
| n/a | |||
| Name | BDBM144672 | ||
| Synonyms: | US8952037, 45 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C21H19N5O2S | ||
| Mol. Mass. | 405.473 | ||
| SMILES | COc1ncccc1-c1nccnc1O[C@@H]1C[C@@H](C1)Nc1nc2ccccc2s1 |r,wU:15.16,17.21,(-8.65,1.54,;-7.32,.77,;-5.98,1.54,;-5.98,3.08,;-4.65,3.85,;-3.32,3.08,;-3.32,1.54,;-4.65,.77,;-4.65,-.77,;-5.98,-1.54,;-5.98,-3.08,;-4.65,-3.85,;-3.32,-3.08,;-3.32,-1.54,;-1.98,-.77,;-.65,-1.54,;-.25,-3.03,;1.24,-2.63,;.84,-1.14,;2.57,-3.4,;3.9,-2.63,;4.07,-1.1,;5.57,-.78,;6.34,.56,;7.88,.56,;8.65,-.78,;7.88,-2.11,;6.34,-2.11,;5.31,-3.26,)| | ||
| Structure | 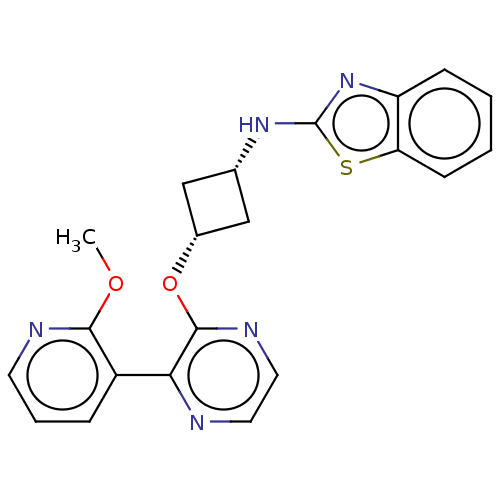
| ||