| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein arginine N-methyltransferase 5 |
|---|
| Ligand | BDBM430627 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | PRMT5 Biochemical Assay |
|---|
| IC50 | 7317±n/a nM |
|---|
| Citation |  Stupple, AE; Walker, SR; Stupple, PA Tetrahydroisoquinolines as PRMT5 inhibitors US Patent US10550096 Publication Date 2/4/2020 Stupple, AE; Walker, SR; Stupple, PA Tetrahydroisoquinolines as PRMT5 inhibitors US Patent US10550096 Publication Date 2/4/2020 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein arginine N-methyltransferase 5 |
|---|
| Name: | Protein arginine N-methyltransferase 5 |
|---|
| Synonyms: | 72 kDa ICln-binding protein | ANM5_HUMAN | HRMT1L5 | Histone-arginine N-methyltransferase PRMT5 | IBP72 | JBP1 | Jak-binding protein 1 | PRMT5 | PRMT5/MEP50 complex | Protein arginine N-methyltransferase 5 (PRMT5) | Protein arginine methyltransferase 5 (PRMT5) | SKB1 | SKB1 homolog | SKB1Hs | Shk1 kinase-binding protein 1 homolog |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 72679.99 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O14744 |
|---|
| Residue: | 637 |
|---|
| Sequence: | MAAMAVGGAGGSRVSSGRDLNCVPEIADTLGAVAKQGFDFLCMPVFHPRFKREFIQEPAK
NRPGPQTRSDLLLSGRDWNTLIVGKLSPWIRPDSKVEKIRRNSEAAMLQELNFGAYLGLP
AFLLPLNQEDNTNLARVLTNHIHTGHHSSMFWMRVPLVAPEDLRDDIIENAPTTHTEEYS
GEEKTWMWWHNFRTLCDYSKRIAVALEIGADLPSNHVIDRWLGEPIKAAILPTSIFLTNK
KGFPVLSKMHQRLIFRLLKLEVQFIITGTNHHSEKEFCSYLQYLEYLSQNRPPPNAYELF
AKGYEDYLQSPLQPLMDNLESQTYEVFEKDPIKYSQYQQAIYKCLLDRVPEEEKDTNVQV
LMVLGAGRGPLVNASLRAAKQADRRIKLYAVEKNPNAVVTLENWQFEEWGSQVTVVSSDM
REWVAPEKADIIVSELLGSFADNELSPECLDGAQHFLKDDGVSIPGEYTSFLAPISSSKL
YNEVRACREKDRDPEAQFEMPYVVRLHNFHQLSAPQPCFTFSHPNRDPMIDNNRYCTLEF
PVEVNTVLHGFAGYFETVLYQDITLSIRPETHSPGMFSWFPILFPIKQPITVREGQTICV
RFWRCSNSKKVWYEWAVTAPVCSAIHNPTGRSYTIGL
|
|
|
|---|
| BDBM430627 |
|---|
| n/a |
|---|
| Name | BDBM430627 |
|---|
| Synonyms: | US10550096, Example 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H21N3O2 |
|---|
| Mol. Mass. | 335.3996 |
|---|
| SMILES | Cc1nn(C[C@@H](O)[C@@H]2Cc3ccccc3CN2)c(=O)c2ccccc12 |r| |
|---|
| Structure | 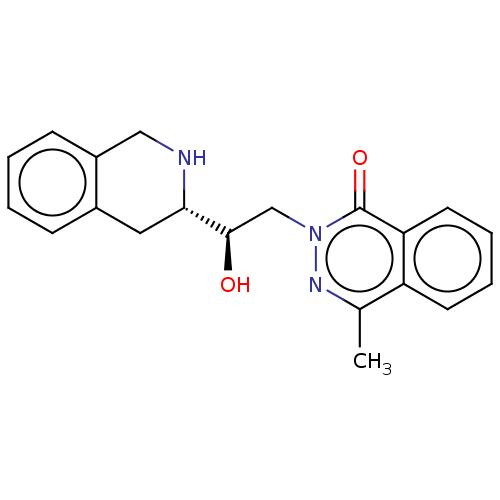
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Stupple, AE; Walker, SR; Stupple, PA Tetrahydroisoquinolines as PRMT5 inhibitors US Patent US10550096 Publication Date 2/4/2020
Stupple, AE; Walker, SR; Stupple, PA Tetrahydroisoquinolines as PRMT5 inhibitors US Patent US10550096 Publication Date 2/4/2020