| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor XII |
|---|
| Ligand | BDBM528108 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Determination of the % Inhibition for FXIIa |
|---|
| IC50 | >4000±n/a nM |
|---|
| Citation |  Davie, RL; Edwards, HJ; Evans, DM; Hodgson, ST; Pethen, SJ; Rooker, DP Pyrazole derivatives as plasma kallikrein inhibitors US Patent US11180484 Publication Date 11/23/2021 Davie, RL; Edwards, HJ; Evans, DM; Hodgson, ST; Pethen, SJ; Rooker, DP Pyrazole derivatives as plasma kallikrein inhibitors US Patent US11180484 Publication Date 11/23/2021 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor XII |
|---|
| Name: | Coagulation factor XII |
|---|
| Synonyms: | Beta-factor XIIa part 1 | Beta-factor XIIa part 2 | Carboxylesterase 2 (intestine, liver) | Coagulation factor XII | Coagulation factor XII (FXII) | Coagulation factor XIIa heavy chain | Coagulation factor XIIa light chain | F12 | FA12_HUMAN | Factor XIIa | Factor XIIa (fXIIa) | HAF | Hageman factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67810.07 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00748 |
|---|
| Residue: | 615 |
|---|
| Sequence: | MRALLLLGFLLVSLESTLSIPPWEAPKEHKYKAEEHTVVLTVTGEPCHFPFQYHRQLYHK
CTHKGRPGPQPWCATTPNFDQDQRWGYCLEPKKVKDHCSKHSPCQKGGTCVNMPSGPHCL
CPQHLTGNHCQKEKCFEPQLLRFFHKNEIWYRTEQAAVARCQCKGPDAHCQRLASQACRT
NPCLHGGRCLEVEGHRLCHCPVGYTGAFCDVDTKASCYDGRGLSYRGLARTTLSGAPCQP
WASEATYRNVTAEQARNWGLGGHAFCRNPDNDIRPWCFVLNRDRLSWEYCDLAQCQTPTQ
AAPPTPVSPRLHVPLMPAQPAPPKPQPTTRTPPQSQTPGALPAKREQPPSLTRNGPLSCG
QRLRKSLSSMTRVVGGLVALRGAHPYIAALYWGHSFCAGSLIAPCWVLTAAHCLQDRPAP
EDLTVVLGQERRNHSCEPCQTLAVRSYRLHEAFSPVSYQHDLALLRLQEDADGSCALLSP
YVQPVCLPSGAARPSETTLCQVAGWGHQFEGAEEYASFLQEAQVPFLSLERCSAPDVHGS
SILPGMLCAGFLEGGTDACQGDSGGPLVCEDQAAERRLTLQGIISWGSGCGDRNKPGVYT
DVAYYLAWIREHTVS
|
|
|
|---|
| BDBM528108 |
|---|
| n/a |
|---|
| Name | BDBM528108 |
|---|
| Synonyms: | N-[(6-carbamoyl-2-fluoro-3-methoxyphenyl)methyl]-3-(methoxymethyl)-1-({4-[(2- oxopyridin-1-yl)methyl]phenyl}methyl)pyrazole-4-carboxamide | US11180484, Example 42 | US11180484, Example 43 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H28FN5O5 |
|---|
| Mol. Mass. | 533.5508 |
|---|
| SMILES | COCc1nn(Cc2ccc(Cn3ccccc3=O)cc2)cc1C(=O)NCc1c(F)c(OC)ccc1C(N)=O |
|---|
| Structure | 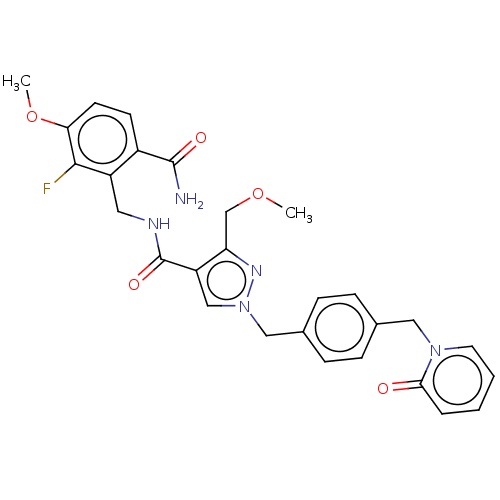
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Davie, RL; Edwards, HJ; Evans, DM; Hodgson, ST; Pethen, SJ; Rooker, DP Pyrazole derivatives as plasma kallikrein inhibitors US Patent US11180484 Publication Date 11/23/2021
Davie, RL; Edwards, HJ; Evans, DM; Hodgson, ST; Pethen, SJ; Rooker, DP Pyrazole derivatives as plasma kallikrein inhibitors US Patent US11180484 Publication Date 11/23/2021