| Reaction Details |
|---|
 | Report a problem with these data |
| Target | POsterior Segregation |
|---|
| Ligand | BDBM52378 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of POS-1 Binding to mex-3-RNA |
|---|
| EC50 | 300000±n/a nM |
|---|
| Citation |  PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of POS-1 Binding to mex-3-RNA PubChem Bioassay(2009)[AID] PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of POS-1 Binding to mex-3-RNA PubChem Bioassay(2009)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| POsterior Segregation |
|---|
| Name: | POsterior Segregation |
|---|
| Synonyms: | POsterior Segregation family member (pos-1) |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 29838.90 |
|---|
| Organism: | Caenorhabditis elegans |
|---|
| Description: | gi_17562800 |
|---|
| Residue: | 264 |
|---|
| Sequence: | MADNDFLSGEAIMVFKKEILDSHSDFTRSLSHQSASPEAYDQENVFSQDFQPFMKQDKET
QNSASQPTSEQSLANRDPCTVPDDLREEMMRQRRKEDAFKTALCDAYKRSQACSYGDQCR
FAHGVHELRLPMNPRGRNHPKYKTVLCDKFSMTGNCKYGTRCQFIHKIVDGNAAKLASGA
HANTSSKSPARNAAAHNHSLFVPQGSTDRSMDLNQSLPIRQSDLVRAFARATRLDVSGYN
STAQLAQYFESQFQRIQQLSSHHH
|
|
|
|---|
| BDBM52378 |
|---|
| n/a |
|---|
| Name | BDBM52378 |
|---|
| Synonyms: | MLS000546183 | N-(2-cyanoethyl)-N-({5-[(1-(4-methylphenyl)-4,6-dioxo-2-thioxotetrahydro-5(2H)-pyrimidinylidene)methyl]-2-furyl}methyl)methanesulfonamide | N-(2-cyanoethyl)-N-[[5-[(E)-[1-(4-methylphenyl)-4,6-bis(oxidanylidene)-2-sulfanylidene-1,3-diazinan-5-ylidene]methyl]furan-2-yl]methyl]methanesulfonamide | N-(2-cyanoethyl)-N-[[5-[(E)-[1-(4-methylphenyl)-4,6-dioxo-2-sulfanylidene-1,3-diazinan-5-ylidene]methyl]-2-furanyl]methyl]methanesulfonamide | N-(2-cyanoethyl)-N-[[5-[(E)-[1-(4-methylphenyl)-4,6-dioxo-2-sulfanylidene-1,3-diazinan-5-ylidene]methyl]furan-2-yl]methyl]methanesulfonamide | N-(2-cyanoethyl)-N-[[5-[(E)-[4,6-diketo-1-(p-tolyl)-2-thioxo-hexahydropyrimidin-5-ylidene]methyl]-2-furyl]methyl]methanesulfonamide | SMR000162492 | cid_1814301 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H20N4O5S2 |
|---|
| Mol. Mass. | 472.537 |
|---|
| SMILES | Cc1ccc(cc1)N1C(=S)NC(=O)C(=Cc2ccc(CN(CCC#N)S(C)(=O)=O)o2)C1=O |w:14.15| |
|---|
| Structure | 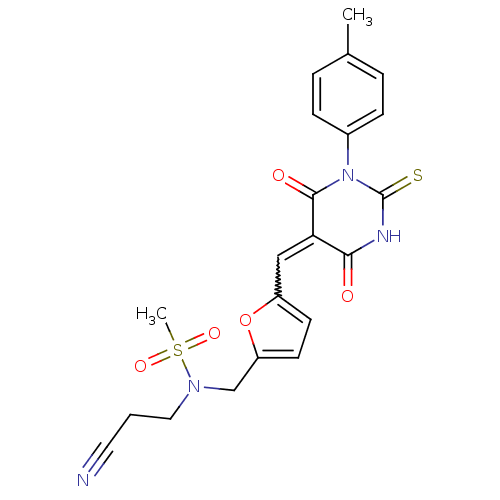
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of POS-1 Binding to mex-3-RNA PubChem Bioassay(2009)[AID]
PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of POS-1 Binding to mex-3-RNA PubChem Bioassay(2009)[AID]