| Reaction Details |
|---|
 | Report a problem with these data |
| Target | DNA topoisomerase 2-beta |
|---|
| Ligand | BDBM22984 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1780771 (CHEMBL4252288) |
|---|
| IC50 | 3500±n/a nM |
|---|
| Citation |  Abdelhaleem, EF; Abdelhameid, MK; Kassab, AE; Kandeel, MM Design and synthesis of thienopyrimidine urea derivatives with potential cytotoxic and pro-apoptotic activity against breast cancer cell line MCF-7. Eur J Med Chem143:1807-1825 (2018) [PubMed] Article Abdelhaleem, EF; Abdelhameid, MK; Kassab, AE; Kandeel, MM Design and synthesis of thienopyrimidine urea derivatives with potential cytotoxic and pro-apoptotic activity against breast cancer cell line MCF-7. Eur J Med Chem143:1807-1825 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| DNA topoisomerase 2-beta |
|---|
| Name: | DNA topoisomerase 2-beta |
|---|
| Synonyms: | DNA topoisomerase II | DNA topoisomerase II beta | TOP2B | TOP2B_HUMAN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 183284.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_51583 |
|---|
| Residue: | 1626 |
|---|
| Sequence: | MAKSGGCGAGAGVGGGNGALTWVTLFDQNNAAKKEESETANKNDSSKKLSVERVYQKKTQ
LEHILLRPDTYIGSVEPLTQFMWVYDEDVGMNCREVTFVPGLYKIFDEILVNAADNKQRD
KNMTCIKVSIDPESNIISIWNNGKGIPVVEHKVEKVYVPALIFGQLLTSSNYDDDEKKVT
GGRNGYGAKLCNIFSTKFTVETACKEYKHSFKQTWMNNMMKTSEAKIKHFDGEDYTCITF
QPDLSKFKMEKLDKDIVALMTRRAYDLAGSCRGVKVMFNGKKLPVNGFRSYVDLYVKDKL
DETGVALKVIHELANERWDVCLTLSEKGFQQISFVNSIATTKGGRHVDYVVDQVVGKLIE
VVKKKNKAGVSVKPFQVKNHIWVFINCLIENPTFDSQTKENMTLQPKSFGSKCQLSEKFF
KAASNCGIVESILNWVKFKAQTQLNKKCSSVKYSKIKGIPKLDDANDAGGKHSLECTLIL
TEGDSAKSLAVSGLGVIGRDRYGVFPLRGKILNVREASHKQIMENAEINNIIKIVGLQYK
KSYDDAESLKTLRYGKIMIMTDQDQDGSHIKGLLINFIHHNWPSLLKHGFLEEFITPIVK
ASKNKQELSFYSIPEFDEWKKHIENQKAWKIKYYKGLGTSTAKEAKEYFADMERHRILFR
YAGPEDDAAITLAFSKKKIDDRKEWLTNFMEDRRQRRLHGLPEQFLYGTATKHLTYNDFI
NKELILFSNSDNERSIPSLVDGFKPGQRKVLFTCFKRNDKREVKVAQLAGSVAEMSAYHH
GEQALMMTIVNLAQNFVGSNNINLLQPIGQFGTRLHGGKDAASPRYIFTMLSTLARLLFP
AVDDNLLKFLYDDNQRVEPEWYIPIIPMVLINGAEGIGTGWACKLPNYDAREIVNNVRRM
LDGLDPHPMLPNYKNFKGTIQELGQNQYAVSGEIFVVDRNTVEITELPVRTWTQVYKEQV
LEPMLNGTDKTPALISDYKEYHTDTTVKFVVKMTEEKLAQAEAAGLHKVFKLQTTLTCNS
MVLFDHMGCLKKYETVQDILKEFFDLRLSYYGLRKEWLVGMLGAESTKLNNQARFILEKI
QGKITIENRSKKDLIQMLVQRGYESDPVKAWKEAQEKAAEEDETQNQHDDSSSDSGTPSG
PDFNYILNMSLWSLTKEKVEELIKQRDAKGREVNDLKRKSPSDLWKEDLAAFVEELDKVE
SQEREDVLAGMSGKAIKGKVGKPKVKKLQLEETMPSPYGRRIIPEITAMKADASKKLLKK
KKGDLDTAAVKVEFDEEFSGAPVEGAGEEALTPSVPINKGPKPKREKKEPGTRVRKTPTS
SGKPSAKKVKKRNPWSDDESKSESDLEETEPVVIPRDSLLRRAAAERPKYTFDFSEEEDD
DADDDDDDNNDLEELKVKASPITNDGEDEFVPSDGLDKDEYTFSPGKSKATPEKSLHDKK
SQDFGNLFSFPSYSQKSEDDSAKFDSNEEDSASVFSPSFGLKQTDKVPSKTVAAKKGKPS
SDTVPKPKRAPKQKKVVEAVNSDSDSEFGIPKKTTTPKGKGRGAKKRKASGSENEGDYNP
GRKTSKTTSKKPKKTSFDQDSDVDIFPSDFPTEPPSLPRTGRARKEVKYFAESDEEEDDV
DFAMFN
|
|
|
|---|
| BDBM22984 |
|---|
| n/a |
|---|
| Name | BDBM22984 |
|---|
| Synonyms: | (8S,10S)-10-{[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy}-6,8,11-trihydroxy-8-(2-hydroxyacetyl)-1-methoxy-5,7,8,9,10,12-hexahydrotetracene-5,12-dione | Adriamycin | Adriblastina | CHEMBL53463 | Doxil | Doxorubicin |
|---|
| Type | Antibiotic |
|---|
| Emp. Form. | C27H29NO11 |
|---|
| Mol. Mass. | 543.5193 |
|---|
| SMILES | COc1cccc2C(=O)c3c(O)c4C[C@](O)(C[C@H](O[C@H]5C[C@H](N)[C@H](O)[C@H](C)O5)c4c(O)c3C(=O)c12)C(=O)CO |r| |
|---|
| Structure | 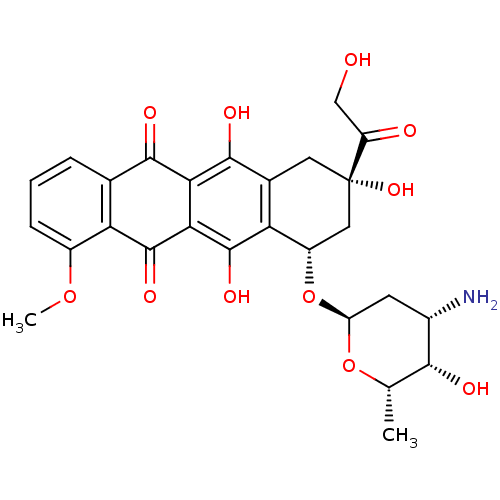
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Abdelhaleem, EF; Abdelhameid, MK; Kassab, AE; Kandeel, MM Design and synthesis of thienopyrimidine urea derivatives with potential cytotoxic and pro-apoptotic activity against breast cancer cell line MCF-7. Eur J Med Chem143:1807-1825 (2018) [PubMed] Article
Abdelhaleem, EF; Abdelhameid, MK; Kassab, AE; Kandeel, MM Design and synthesis of thienopyrimidine urea derivatives with potential cytotoxic and pro-apoptotic activity against breast cancer cell line MCF-7. Eur J Med Chem143:1807-1825 (2018) [PubMed] Article