| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Inosine-5'-monophosphate dehydrogenase |
|---|
| Ligand | BDBM31072 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1797185 (CHEMBL4269302) |
|---|
| IC50 | 370±n/a nM |
|---|
| Citation |  Kummari, LK; Butler, MS; Furlong, E; Blundell, R; Nouwens, A; Silva, AB; Kappler, U; Fraser, JA; Kobe, B; Cooper, MA; Robertson, AAB Antifungal benzo[b]thiophene 1,1-dioxide IMPDH inhibitors exhibit pan-assay interference (PAINS) profiles. Bioorg Med Chem26:5408-5419 (2018) [PubMed] Article Kummari, LK; Butler, MS; Furlong, E; Blundell, R; Nouwens, A; Silva, AB; Kappler, U; Fraser, JA; Kobe, B; Cooper, MA; Robertson, AAB Antifungal benzo[b]thiophene 1,1-dioxide IMPDH inhibitors exhibit pan-assay interference (PAINS) profiles. Bioorg Med Chem26:5408-5419 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Inosine-5'-monophosphate dehydrogenase |
|---|
| Name: | Inosine-5'-monophosphate dehydrogenase |
|---|
| Synonyms: | 1.1.1.205 | IMDH_CRYNJ | IMP dehydrogenase | IMPD | IMPDH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57901.81 |
|---|
| Organism: | Cryptococcus neoformans var. neoformans serotype D (strain JEC21 /ATCC MYA-565) (Filobasidiella neoformans) |
|---|
| Description: | ChEMBL_117850 |
|---|
| Residue: | 544 |
|---|
| Sequence: | MADTNPNAPPRSDSLLNPADALKYLEEYPRGDGLSLQELMDSRKNGGLTYNDFLVLPGHI
SFPASDVSLQSRATKNIVLNTPFLSSPMDTVTEDRMAIALALHGGLGIIHHNCSAEEQAA
MVRRVKKYENGFITDPLCLGPDATVGDVLEIKAKFGFCGVPITETGAPNSKLLGIVTGRD
VQFQDAETPIKSVMTTEVVTGSSPITLEKANSLLRETKKGKLPIVDSNGHLVSLVARSDL
LKNQNYPYASKVPESKQLYCGAAIGTRPGDKDRLKLLAEAGLDVVVLDSSQGDSVYQIEF
IKWIKQTYPKIEIIAGNVVTREQAAQLIAAGADGLRIGMGSGSICITQEVMAVGRPQGTA
VYAVAEFASRFGIPCIADGGIGNIGHIAKALALGASAVMMGGLLAGTTESPGEYFYHEGK
RVKVYRGMGSIEAMEHTQRGSASGKRSILNLDNAATARYFSEADAVKVAQGVSGDVADKG
SINKFVPYLFTGLQHSFQDAGVKSVSELHSCARSGSLRFELRTASAQLEGGVHGLNSYTK
RLFA
|
|
|
|---|
| BDBM31072 |
|---|
| n/a |
|---|
| Name | BDBM31072 |
|---|
| Synonyms: | 2-[(1,1-dioxido-1-benzothien-3-yl)thio]-5-phenyl-1,3,4-oxadiazole | 3-[(5-phenyl-1,3,4-oxadiazol-2-yl)sulfanyl]-1-benzothiophene 1,1-dioxide | 3-[(5-phenyl-1,3,4-oxadiazol-2-yl)thio]-1-benzothiophene 1,1-dioxide | 3-[(5-phenyl-1,3,4-oxadiazol-2-yl)thio]benzothiophene 1,1-dioxide | MLS000088644 | SMR000072650 | cid_890649 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H10N2O3S2 |
|---|
| Mol. Mass. | 342.392 |
|---|
| SMILES | O=S1(=O)C=C(Sc2nnc(o2)-c2ccccc2)c2ccccc12 |t:3| |
|---|
| Structure | 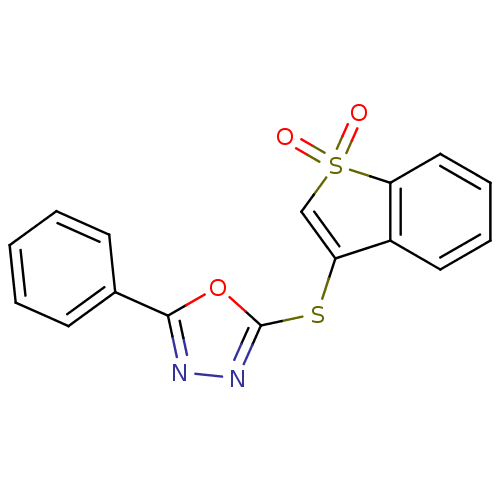
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kummari, LK; Butler, MS; Furlong, E; Blundell, R; Nouwens, A; Silva, AB; Kappler, U; Fraser, JA; Kobe, B; Cooper, MA; Robertson, AAB Antifungal benzo[b]thiophene 1,1-dioxide IMPDH inhibitors exhibit pan-assay interference (PAINS) profiles. Bioorg Med Chem26:5408-5419 (2018) [PubMed] Article
Kummari, LK; Butler, MS; Furlong, E; Blundell, R; Nouwens, A; Silva, AB; Kappler, U; Fraser, JA; Kobe, B; Cooper, MA; Robertson, AAB Antifungal benzo[b]thiophene 1,1-dioxide IMPDH inhibitors exhibit pan-assay interference (PAINS) profiles. Bioorg Med Chem26:5408-5419 (2018) [PubMed] Article