| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2C |
|---|
| Ligand | BDBM50176019 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_446511 (CHEMBL895623) |
|---|
| Ki | 200±n/a nM |
|---|
| Citation |  Lange, JH; Reinders, JH; Tolboom, JT; Glennon, JC; Coolen, HK; Kruse, CG Principal component analysis differentiates the receptor binding profiles of three antipsychotic drug candidates from current antipsychotic drugs. J Med Chem50:5103-8 (2007) [PubMed] Article Lange, JH; Reinders, JH; Tolboom, JT; Glennon, JC; Coolen, HK; Kruse, CG Principal component analysis differentiates the receptor binding profiles of three antipsychotic drug candidates from current antipsychotic drugs. J Med Chem50:5103-8 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2C |
|---|
| Name: | 5-hydroxytryptamine receptor 2C |
|---|
| Synonyms: | 5-HT-1C | 5-HT-2C | 5-HT1C | 5-HT2C | 5-HT2C-INI | 5-HT2c VGI | 5-HTR2C | 5-hydroxytryptamine receptor 1C | 5-hydroxytryptamine receptor 2C (5-HT-2C) | 5-hydroxytryptamine receptor 2C (5HT-2C) | 5HT-1C | 5HT2C_HUMAN | HTR1C | HTR2C | Serotonin (5-HT3) receptor | Serotonin 2c (5-HT2c) receptor | Serotonin Receptor 2C |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 51836.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28335 |
|---|
| Residue: | 458 |
|---|
| Sequence: | MVNLRNAVHSFLVHLIGLLVWQSDISVSPVAAIVTDIFNTSDGGRFKFPDGVQNWPALSI
VIIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYVW
PLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVWA
ISIGVSVPIPVIGLRDEEKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYCLTIYV
LRRQALMLLHGHTEEPPGLSLDFLKCCKRNTAEEENSANPNQDQNARRRKKKERRPRGTM
QAINNERKASKVLGIVFFVFLIMWCPFFITNILSVLCEKSCNQKLMEKLLNVFVWIGYVC
SGINPLVYTLFNKIYRRAFSNYLRCNYKVEKKPPVRQIPRVAATALSGRELNVNIYRHTN
EPVIEKASDNEPGIEMQVENLELPVNPSSVVSERISSV
|
|
|
|---|
| BDBM50176019 |
|---|
| n/a |
|---|
| Name | BDBM50176019 |
|---|
| Synonyms: | (R)-8-(4-(3-(5-fluoro-1H-indol-3-yl)propyl)piperazin-1-yl)-2-methyl-2H-benzo[b][1,4]oxazin-3(4H)-one | (R)-8-{4-[3-(5-Fluoro-1H-indol-3-yl)-propyl]-piperazin-1-yl}-2-methyl-4H-benzo[1,4]oxazin-3-one | CHEMBL196514 | SLV-314 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H27FN4O2 |
|---|
| Mol. Mass. | 422.4952 |
|---|
| SMILES | C[C@H]1Oc2c(NC1=O)cccc2N1CCN(CCCc2c[nH]c3ccc(F)cc23)CC1 |
|---|
| Structure | 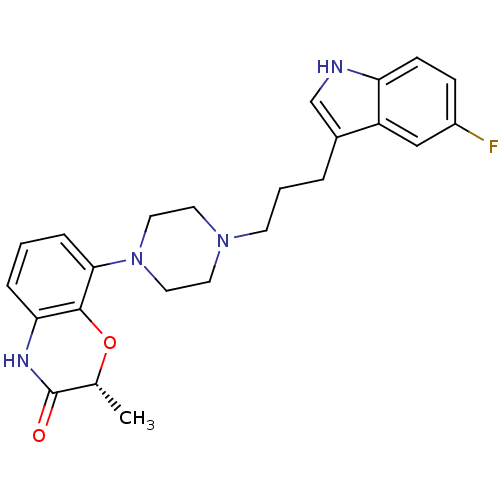
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lange, JH; Reinders, JH; Tolboom, JT; Glennon, JC; Coolen, HK; Kruse, CG Principal component analysis differentiates the receptor binding profiles of three antipsychotic drug candidates from current antipsychotic drugs. J Med Chem50:5103-8 (2007) [PubMed] Article
Lange, JH; Reinders, JH; Tolboom, JT; Glennon, JC; Coolen, HK; Kruse, CG Principal component analysis differentiates the receptor binding profiles of three antipsychotic drug candidates from current antipsychotic drugs. J Med Chem50:5103-8 (2007) [PubMed] Article